Table of Contents
Emerging role of cellular senescence in peritumoral microenvironment on tumor progression
Accumulating evidence has indicated that the normal tissue adjacent to tumor (NAT) is distinct from both healthy and tumor tissues. It is suggested that the crosstalk between NAT and tumor tissues helps shape the tumor microenvironment and promotes cancer ...
More.Accumulating evidence has indicated that the normal tissue adjacent to tumor (NAT) is distinct from both healthy and tumor tissues. It is suggested that the crosstalk between NAT and tumor tissues helps shape the tumor microenvironment and promotes cancer progression, but the molecular and cellular evidence for this crosstalk is scarce. In this perspective, we propose that NAT tissue constitutes a unique “peritumoral microenvironment (Peri-TME)” mostly conditioned by the tumor. Furthermore, cellular senescence is identified as a key characteristic of Peri-TME, which accelerates tumor growth, as illustrated in our recent studies in colorectal cancer (CRC). Finally, strategies to target the senescent Peri-TME may represent an effective means to disrupt the vicious interaction between Peri-TME and Tumor and enhance therapeutic efficacy.
Less.Mantang Zhou, ... Dongwang Yan
DOI:https://doi.org/10.70401/Geromedicine.2026.0021 - April 24, 2026
Encouraging a move toward precision geromedicine
Aging is a heterogeneous, multi-system process driven by the interplay between accumulating molecular damage and the progressive erosion of resilience. While damage accumulates in a ubiquitous and homogeneous fashion, resilience is finite and unequally ...
More.Aging is a heterogeneous, multi-system process driven by the interplay between accumulating molecular damage and the progressive erosion of resilience. While damage accumulates in a ubiquitous and homogeneous fashion, resilience is finite and unequally distributed across physiological systems and individuals, yielding distinct biological trajectories that diverge early in life, giving rise to the individual-specific decline in physiological function and the manifestation of a diverse spectrum of organ-specific diseases, and converge only when multisystem dysregulation overwhelms compensatory capacity. Early deviations in mitochondrial function, proteostasis, inflammation, and metabolic regulation often remain clinically silent, detectable only through gerodiagnostics, longitudinal, sensitive biomarkers of aging. Yet most biomarkers were developed to detect disease rather than quantify aging biology, and their mechanistic specificity declines with advancing multimorbidity. Precision geromedicine therefore requires the integration of gerodiagnostics that capture system-level resilience and stress responsiveness with measures of functional reserve, behavior, physiology, and the exposome, enabling the identification of individualized aging trajectories and the biological pathways that drive them. This combined approach clarifies causal pathways, enables earlier detection of vulnerability and supports individualized gerointerventions that modify aging trajectories rather than specifically but narrowly focusing on individual age-related diseases.
Less.Luigi Ferrucci, ... Guido Kroemer
DOI:https://doi.org/10.70401/Geromedicine.2026.0020 - April 23, 2026
The role of glia autophagy in CNS homeostasis, ageing and disease
Autophagy is a fundamental catabolic process that is critical for maintaining cellular homeostasis and protein quality control in the central nervous system (CNS). While neuronal autophagy has been extensively characterized, growing evidence highlights ...
More.Autophagy is a fundamental catabolic process that is critical for maintaining cellular homeostasis and protein quality control in the central nervous system (CNS). While neuronal autophagy has been extensively characterized, growing evidence highlights the indispensable roles of glial autophagy, specifically in astrocytes, oligodendrocytes and microglia, in CNS physiology and pathology. These glial populations employ the autophagic machinery to regulate distinct but interconnected functions: astrocytes manage metabolic support and glutamate homeostasis; oligodendrocytes rely on autophagic flux for differentiation and myelin maintenance; and microglia employ specific pathways, such as LC3-associated phagocytosis, to orchestrate immune surveillance and inflammasome regulation. Impairment of glial autophagy has been implicated in non-cell-autonomous neurodegeneration, leading to excitotoxicity, myelin damage, the emergence of senescence-associated secretory phenotypes, and persistent neuroinflammation. Dysregulation of autophagic pathways during ageing has also been implicated in the pathogenesis of multiple neurodegenerative diseases, including Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, and amyotrophic lateral sclerosis. In this review we summarize the cell-type-specific molecular mechanisms of autophagy in glia, and delineate their role in the clearance of pathogenic aggregates such as β-amyloid, α-synuclein, and mutant huntingtin. A deeper understanding of the spatiotemporal dynamics of glial autophagy and associated intercellular crosstalk is essential to fully elucidate the complex etiology of age-associated neurodegenerative conditions.
Less.Onur Çakıcı, Nektarios Tavernarakis
DOI:https://doi.org/10.70401/Geromedicine.2026.0019 - March 23, 2026
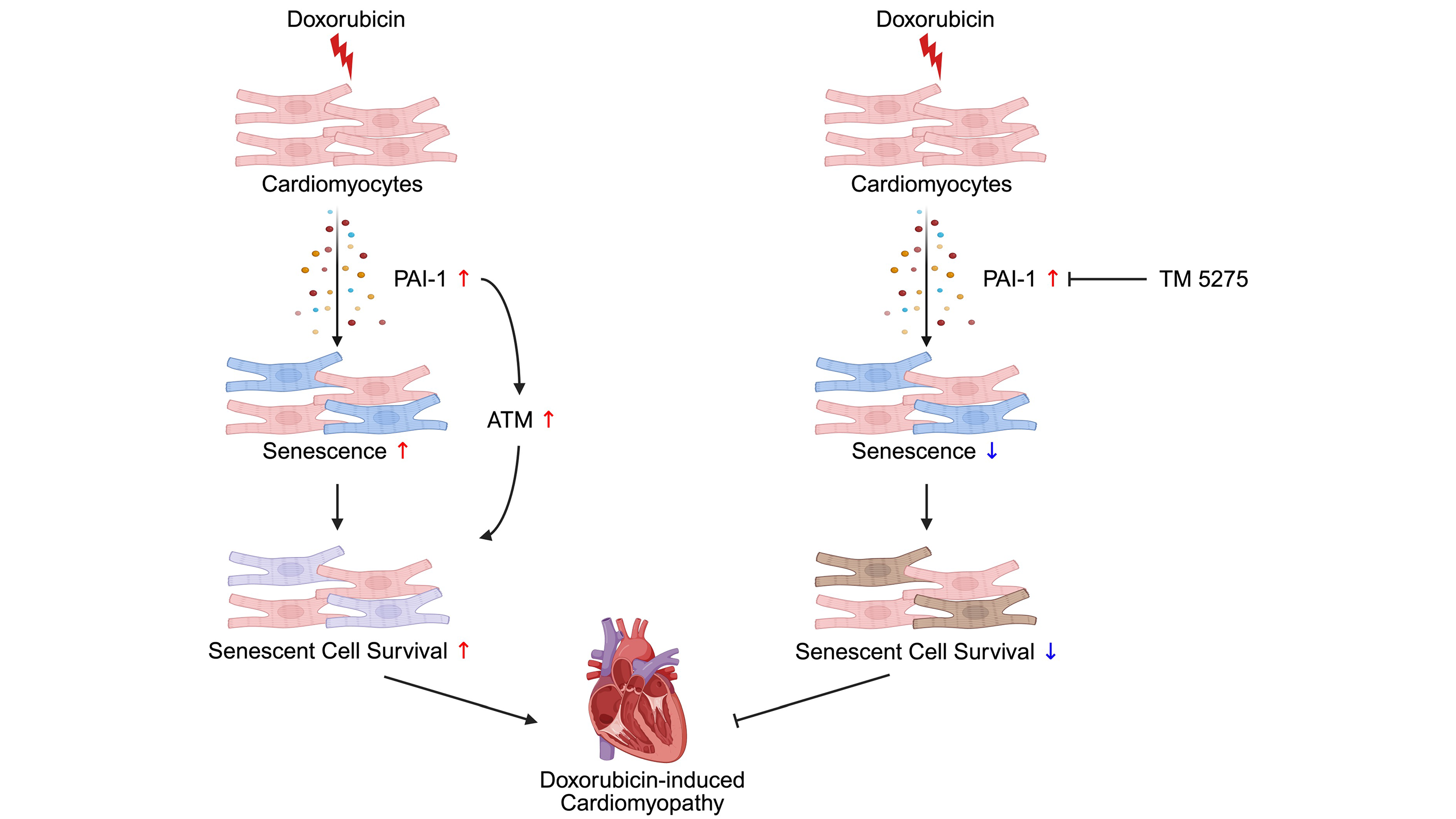
Inhibition of PAI-1 shifts cardiomyocyte fate from senescence toward apoptosis and mitigates doxorubicin-induced cardiotoxicity
Aims: Doxorubicin (Dox) is an effective chemotherapeutic agent, but its clinical use is limited by cardiotoxicity. Cellular senescence contributes to Dox-induced cardiac dysfunction; however, the underlying molecular mechanism mediating the ...
More.Aims: Doxorubicin (Dox) is an effective chemotherapeutic agent, but its clinical use is limited by cardiotoxicity. Cellular senescence contributes to Dox-induced cardiac dysfunction; however, the underlying molecular mechanism mediating the effect of senescence remains poorly understood. This study aimed to identify senescence-associated factors secreted from cardiomyocytes in Dox-treated hearts and define their functional significance in Dox-induced cardiotoxicity.
Methods: Mice with cardiomyocyte-specific expression of the endoplasmic reticulum BioID secretome profiling system were used to identify Dox-induced secreted factors. Functional analyses were performed in neonatal rat ventricular myocytes (NRVMs). The effects of plasminogen activator inhibitor-1 (PAI-1) inhibition were evaluated in Dox-treated mice by assessing senescence markers, apoptotic responses, and cardiac structure and function. p21High-tdTomato reporter mice were used to examine the fate of senescent cardiomyocytes in vivo.
Results: PAI-1 was identified as a major component of the senescence-associated secretory phenotype and was robustly upregulated in Dox-treated cardiomyocytes. In NRVMs, PAI-1 promoted senescence and maintained the senescent phenotype, in part by conferring resistance to apoptosis. Pharmacological inhibition of PAI-1 reduced senescence markers, enhanced apoptotic responses, and preserved cardiac structure and function in Dox-treated mice. Fate mapping analyses with p21High-tdTomato mice revealed that PAI-1 inhibition decreased the number of p21High senescent cardiomyocytes in Dox-treated hearts. Notably, PAI-1 inhibition did not attenuate Dox cytotoxicity in EO771 murine breast cancer cells.
Conclusion: PAI-1 is a key mediator of Dox-induced cardiac dysfunction. PAI-1 inhibition shifts the fate of cardiomyocytes from senescence toward apoptosis and preserves cardiac structure and function without compromising the antitumor function of Dox, highlighting PAI-1 as a potential therapeutic target for chemotherapy-associated cardiotoxicity.
Less.Yuka Shiheido-Watanabe, ... Junichi Sadoshima
DOI:https://doi.org/10.70401/Geromedicine.2026.0018 - March 17, 2026
Nucleolar expansion: A biomolecular condensate mortality timer
The nucleolus, the largest membraneless organelle in the cell, is a biomolecular condensate that houses ribosomal DNA (rDNA), facilitates ribosomal subunit assembly, and serves as a dynamic reservoir for numerous unrelated proteins. Aging across eukaryotic ...
More.The nucleolus, the largest membraneless organelle in the cell, is a biomolecular condensate that houses ribosomal DNA (rDNA), facilitates ribosomal subunit assembly, and serves as a dynamic reservoir for numerous unrelated proteins. Aging across eukaryotic species is accompanied by nucleolar expansion, raising the question of whether it is a correlate of aging or a driver of cellular aging. Recent studies suggest that nucleolar expansion may drive aging and this may result from age-associated changes in the biophysical properties of the nucleolus. Emerging evidence points to age-driven biophysical changes in the nucleolar condensate, including shifts in size, dynamics, and viscoelasticity, which may occur gradually or through transitions from a liquid-like state to denser gel-like, and in some contexts amyloid-like, assemblies. These transitions remodel two core condensate properties: compartmentalization and partitioning, with consequences for ribosome biogenesis and rDNA stability. Here, we review recent literature on age-driven changes in nucleolar condensation and discuss how these changes may influence nucleolar function and longevity.
Less.J. Ignacio Gutierrez, Jessica K. Tyler
DOI:https://doi.org/10.70401/Geromedicine.2026.0017 - March 11, 2026
Stress granules: Emerging regulators of reproductive aging
Reproductive aging progressively impairs fertility and contributes to broader systemic decline. Stress granules (SGs), transient membraneless ribonucleoprotein assemblies formed during cellular stress, have recently emerged as important regulators ...
More.Reproductive aging progressively impairs fertility and contributes to broader systemic decline. Stress granules (SGs), transient membraneless ribonucleoprotein assemblies formed during cellular stress, have recently emerged as important regulators of gonadal homeostasis. Their function is highly context dependent: properly resolved SGs promote cellular adaptation and survival, whereas persistent SGs disrupt proteostasis and trigger cell death pathways. In the testis, transient SGs protect germ cells under stress; however, persistent SG accumulation activates necroptosis through the ZBP1-RIPK3 axis, a pathway implicated in human non-obstructive azoospermia and testicular aging. In the ovary, defective autophagic clearance leads to pathological SG persistence in granulosa cells, while several SG-associated proteins are indispensable for normal oogenesis. Together, these findings indicate that dysregulated SG dynamics, particularly impaired clearance, represent a convergent mechanism linking cellular stress responses to reproductive decline. Despite these advances, critical gaps remain. The cell-type-specific regulation of SG assembly and disassembly within the gonad is not fully defined, and the molecular mechanisms by which persistent SGs drive tissue-level aging require clarification. Addressing these questions will refine our understanding of reproductive aging and its mechanistic connection to proteostatic imbalance.
Less.Nianyu Li, ... Xue Jiao
DOI:https://doi.org/10.70401/Geromedicine.2026.0016 - March 06, 2026