Table of Contents
The caspase-2 paradox in liver polyploidy and cancer risk
Caspase-2 is a key genome-surveillance protease that functions to kill or arrest cells with abnormal chromosome content, via PIDDosome-dependent and -independent mechanisms. However, this surveillance function presents a biological paradox in the liver, ...
More.Caspase-2 is a key genome-surveillance protease that functions to kill or arrest cells with abnormal chromosome content, via PIDDosome-dependent and -independent mechanisms. However, this surveillance function presents a biological paradox in the liver, where physiological polyploidy, is essential for organogenesis, genomic buffering and adaptive stress responses to maintain liver homeostasis. While short-term caspase-2 loss promotes adaptive polyploidy, our recent work demonstrates that prolonged caspase-2 deficiency drives pathogenic hyperploidy, fuelling chronic inflammation, and increased age-associated hepatocellular carcinoma in mice. These findings underscore the need for tight ploidy control in the maintenance of liver homeostasis. They also highlight potential risks for therapeutic targeting of caspase-2 in fatty liver disease and suggest that pathways governing lipid metabolism also influence long-term tumour surveillance mechanisms. This perspective discusses caspase-2 as a guardian of hepatic genome integrity, and the dual, context-dependent roles of polyploidy in liver physiology and metabolic liver disease.
Less.Loretta Dorstyn, Sharad Kumar
DOI:https://doi.org/10.70401/EXO.2026.0006 - April 15, 2026
Human iPSC-derived macrophages for studying intrinsic and extrinsic factors in cystic fibrosis
Background: Cystic fibrosis (CF) is a progressive genetic disease characterized by defective ion transport, mucus accumulation, chronic infection, and inflammation that drive airway damage and ultimately end-stage lung failure. Previous studies ...
More.Background: Cystic fibrosis (CF) is a progressive genetic disease characterized by defective ion transport, mucus accumulation, chronic infection, and inflammation that drive airway damage and ultimately end-stage lung failure. Previous studies show that high levels of proteolytic enzymes in the sputum of CF patients correlate with declining lung function, but the related effects on distal lung extracellular matrix (ECM) and immune responses are unclear.
Methods: To address this gap, induced pluripotent stem cell (iPSC) lines from healthy donors and CF patients were differentiated into macrophages, and stimulated with lipopolysaccharide (LPS) to compare their inflammatory responses. Bulk RNA sequencing, functional assays, and secreted protein profiling revealed key differences between healthy and CF-derived macrophages, providing insight into how these cells may contribute to inflammatory responses in CF patients. Further, human lung ECM from distal CF lung tissue was isolated, used to generate ECM biomaterials, and combined with iPSC-derived macrophages from healthy and CF donors in vitro. Macrophage phenotype was evaluated through cytokine profiling and RNA sequencing.
Results: CF macrophage inflammation was dysregulated, with elevated baseline IL-8, IL-18, and MCP-1 expression, and a blunted inflammatory response to CF ECM compared to healthy macrophages. By using CF ECM and healthy macrophages, we characterized how healthy cells may be altered in a persistent CF milieu after anticipated CFTR modulator therapy.
Conclusion: These findings reveal altered innate immune behavior in CF and demonstrate the utility of iPSC-derived macrophages for modeling extrinsic immune-ECM interactions in disease.
Less.Daniel Naveed Tavakol, ... Gordana Vunjak-Novakovic
DOI:https://doi.org/10.70401/EXO.2026.0005 - April 10, 2026
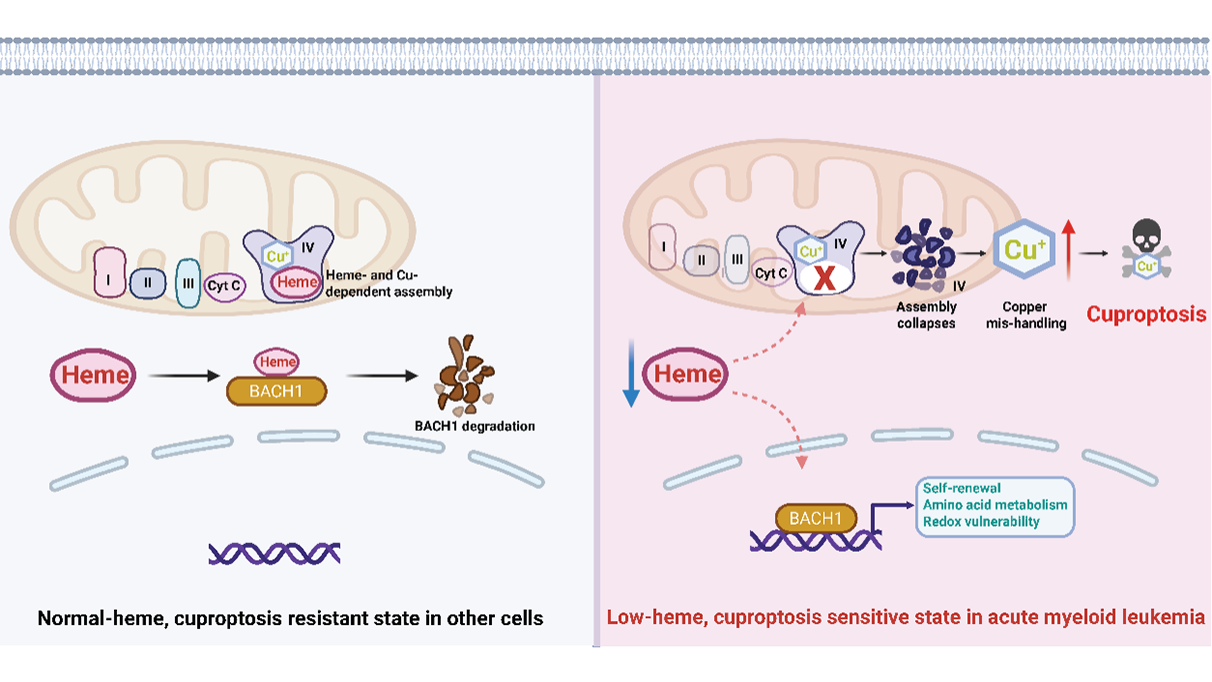
Heme, copper, and a new way to kill cancer cells
Heme homeostasis influences mitochondrial metabolism and leukemia stem cell biology in acute myeloid leukemia. Lewis et al. uncover a surprising metabolic vulnerability in acute myeloid leukemia: suppression of heme biosynthesis primes leukemic ...
More.Heme homeostasis influences mitochondrial metabolism and leukemia stem cell biology in acute myeloid leukemia. Lewis et al. uncover a surprising metabolic vulnerability in acute myeloid leukemia: suppression of heme biosynthesis primes leukemic cells for cuproptosis, a form of copper-dependent cell death. By linking heme depletion to mitochondrial cytochrome c oxidase (Complex IV) dysfunction, copper accumulation, and cuproptosis, the study integrates transcriptional regulation, mitochondrial metabolism, and metal homeostasis into a unified framework for selective cancer cell killing.
Less.Xi Zhao, ... Boyi Gan
DOI:https://doi.org/10.70401/EXO.2026.0004 - March 19, 2026
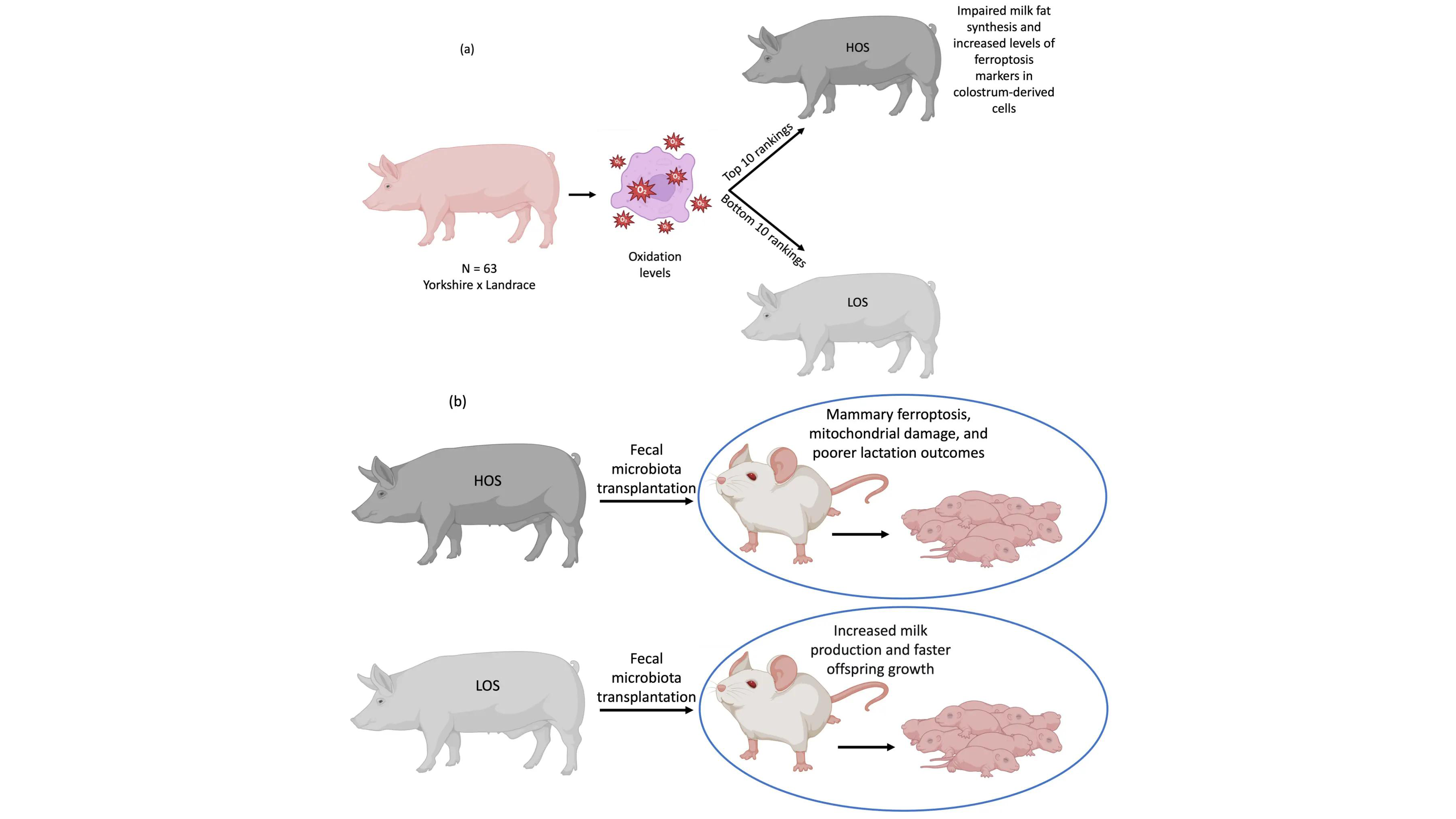
Gut microbial extracellular vesicles as lipid carriers: An emerging paradigm for organ protection and ferroptosis prevention
Ferroptosis is an iron-dependent form of regulated cell death, driven by the extensive peroxidation of cellular membrane phospholipids, particularly those enriched with oxidation-sensitive polyunsaturated fatty acids. Given its role in diverse pathologies, ...
More.Ferroptosis is an iron-dependent form of regulated cell death, driven by the extensive peroxidation of cellular membrane phospholipids, particularly those enriched with oxidation-sensitive polyunsaturated fatty acids. Given its role in diverse pathologies, ferroptosis inhibition represents a compelling therapeutic target. Among the strategies being explored, modulating cellular membrane lipid composition through exogenous supplementation with less oxidizable fatty acids, such as monounsaturated fatty acids, has gained significant attention. Nevertheless, the influence of endogenous regulators on membrane lipid dynamics and ferroptosis susceptibility is not yet fully elucidated and represents a fertile frontier for discovery. While the gut microbiota is well established as a systemic regulator of host physiology, its potential role in modulating membrane lipid composition and ferroptosis susceptibility remains largely unexplored. This Perspective opens by examining the study by Zhang et al., which suggests that bacterial extracellular vesicles (BEVs) from the gut commensal Lactobacillus amylovorus deliver oleic acid to the mammary gland. This mechanism suppresses ferroptosis and helps sustain lactation in mice under oxidative stress. The work provides a proof-of-concept for BEVs as endogenous lipid delivery vectors that may modulate ferroptosis susceptibility across different organs. Building on these findings, this Perspective critically evaluates the conceptual advance represented by Zhang et al. and integrates it with the broader literature and future scientific opportunities. Specifically, the Perspective dissects the mechanistic underpinnings of this pathway within the context of extracellular vesicle biology and inter-organ lipid trafficking. It also maps the unresolved questions poised to shape the future of the field and examines the key translational hurdles that must be overcome to harness BEV-mediated lipid delivery for therapeutic benefit.
Less.Marcelo Farina
DOI:https://doi.org/10.70401/EXO.2026.0003 - March 06, 2026
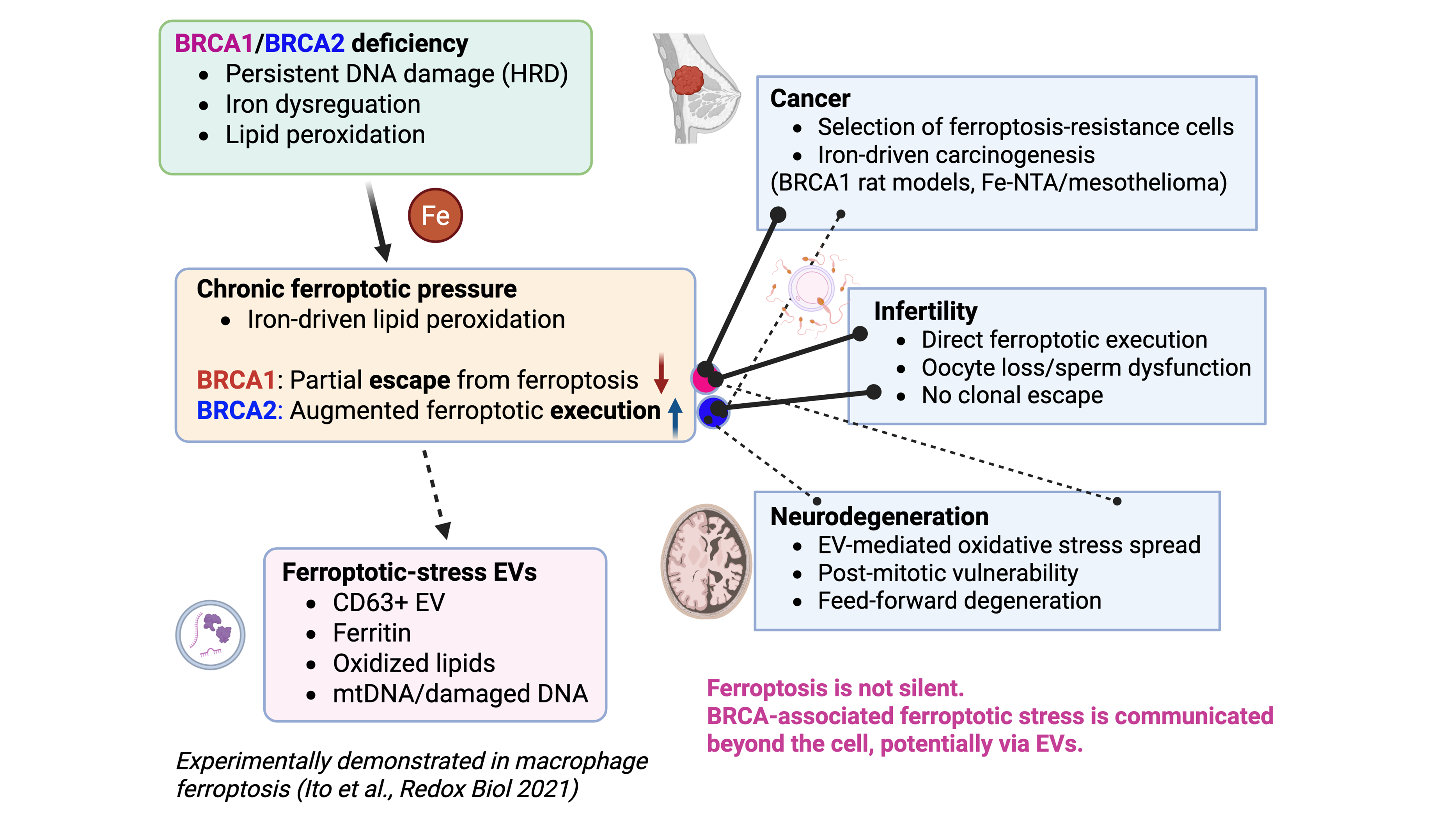
Ferroptosis in BRCA-associated disorders: Extracellular vesicles as potential messengers beyond the cell
BRCA1 and BRCA2 deficiencies are classically defined by impaired homologous recombination–mediated DNA repair; however, their pathological consequences extend far beyond cell-autonomous genomic instability. Accumulating evidence indicates that ...
More.BRCA1 and BRCA2 deficiencies are classically defined by impaired homologous recombination–mediated DNA repair; however, their pathological consequences extend far beyond cell-autonomous genomic instability. Accumulating evidence indicates that BRCA deficiency is accompanied by iron dysregulation and persistent lipid peroxidation, placing cells under chronic ferroptotic pressure. Studies using BRCA1/2 rat models demonstrate that ferroptosis functions as a decisive biological checkpoint with gene-specific outcomes. Under BRCA1 haploinsufficiency, iron-driven oxidative stress accelerates carcinogenesis by selecting for ferroptosis-resistant clones, whereas BRCA2 haploinsufficiency enhances ferroptotic execution, thereby preventing iron-induced cancer promotion. In contrast, reproductive tissues lacking adaptive escape capacity manifest BRCA deficiency as a direct ferroptosis-driven cellular loss, resulting in male and female infertility. Importantly, ferroptosis is not a silent, cell-confined event. Experimental evidence from asbestos-induced carcinogenesis demonstrates that macrophages undergoing ferroptosis after asbestos phagocytosis release CD63-positive, ferritin-containing extracellular vesicles (EVs) that induce oxidative stress in recipient mesothelial cells, establishing EVs as active mediators of ferroptotic stress propagation. We propose that BRCA deficiency generates a state of ferroptotic priming in which oxidized lipids, iron-related factors, and nucleic acids are disseminated via EVs, thereby shaping tissue- and organ-level pathology. From an evolutionary perspective, the persistence of pathogenic BRCA variants may reflect adaptive advantages conferred by haploinsufficiency in iron-limited, short-lived ancestral environments; under modern conditions of iron abundance and extended lifespan, this once-adaptive state becomes maladaptive, predisposing carriers to cancer and degenerative disorders beyond the cell.
Less.Shinya Toyokuni, ... Yashiro Motooka
DOI:https://doi.org/10.70401/EXO.2026.0002 - February 14, 2026