Abstract
Paramorphism effect could be an effective strategy to design efficient catalysts, but has been rarely explored. In this study, to achieve efficient catalysts for elimination of NO and CO together, Ce2Sn2O7 pyrochlore and Ce2Sn2O8+x solid solution paramorphs were purposely synthesized to support FeOx. It is found that Fe/Ce2Sn2O7 displays better reaction performance than Fe/Ce2Sn2O8+x. H2-TPR results have demonstrated that the dispersed FeOx has differed interaction with the two types of supports. Electron paramagnetic resonance (EPR) and density functional theory (DFT) calculation have testified that it is easier to generate surface vacancies on Fe/Ce2Sn2O7 than on Fe/Ce2Sn2O8+x, thus forming more abundant active oxygen sites. Furthermore, the total number of Lewis and Brønsted sites on Fe/Ce2Sn2O7 is larger. In addition, reactive NH4+ linked to Brønsted acidic sites and bridge nitrite are uniquely formed on Fe/Ce2Sn2O7, thus leading to its much better performance than on Fe/Ce2Sn2O8+x. Notably, Fe/Ce2Sn2O7 also exhibits better sulfur and water tolerance. On both catalysts, the NH3-selective catalytic reduction (NH3-SCR) reaction obeys a Langmuir-Hinshelwood pathway, while the CO oxidation follows a Mars-van Krevelen mechanism. In summary, a paramorphism effect is obviously observed, which could give people some new thoughts to design high-performance catalysts.
Keywords
1. Introduction
NOx and CO, which are mainly generated by the burning of fossil fuels in stationary and mobile sources, have resulted in a mass of air pollution problems and destruction of the ozone layer. Thus, the elimination of NOx and CO is extremely important. Selective catalytic reduction of NOx by NH3 (NH3-SCR) is a mainstream de-NOx technology for cleaning exhausts from diesel engines and power plants[1-3]. At present, V2O5-WO3/TiO2 catalyst has been commercialized and widely used in these de-NOx processes. However, the biotoxicity of V2O5, weak resistance to SO2 deactivation, and the narrow operation temperature window have driven people to develop more efficient and environmentally benign catalysts with lower costs[4,5]. At present, CO removing mainly depends on oxidation. Obtaining high-efficiency to remove simultaneously NO and CO holds promising industrial prospects. However, it is still of great necessity to design high-performance catalysts to accomplish this process successfully.
So far, quite several kinds of catalysts have shown considerable reaction performance in conducting NH3-SCR and CO oxidation together[6-9]. Ceria-based catalysts, usually with excellent oxygen storage ability, have drawn a lot of attention[10,11]. It was reported that the NH3-SCR performance on catalysts containing CeO2 can be improved by doping the CeO2 matrix with a secondary cation, or loading other metal oxides onto it[12]. Yang et al. synthesized MnOx-CeO2 catalysts with several methods, and found that a MnOx(0.3)-CeO2 catalyst fabricated with citric acid and calcined at 650 °C exhibited the optimum reaction performance, which achieves nearly 100% NO conversion at 120 °C due to the presence of highly dispersed MnOx species, as well as more abundant active oxygen species[13]. Liu et al. prepared Ce-Sn mixed oxide catalysts for NH3-SCR and found that the interaction between Ce and Sn improved the redox properties and the surface Lewis acidity, which can effectively activate NH3 molecules, thus enhancing the NH3-SCR performance[14]. Moreover, Liu et al. found that a CeO2-SnO2 catalyst fabricated by co-precipitation displayed high activity in 250-400 °C with over 90 % NO conversion and good SO2 tolerance, due to the high dispersion of SnO2 and the formation of a solid solution structure[15]. By introducing acid oxides (W or Nb) into CeO2-SnO2 composite oxides, the acidity can be further increased to improve the NH3-SCR performance, and the tolerance to water vapor and sulfur deactivation[16-19].
In our former studies, SnO2/Beta catalysts with varied SnO2 contents were synthesized for NOx-SCR by propylene based on the monolayer dispersion theory. On a 15% SnO2/Beta sample, which is near the monolayer dispersion capacity, SnO2 is in a sub-monolayer form, thereby forming more surface defects, active O2- and acid sites, which are beneficial to the C3H6-SCR performance[20]. Moreover, we also investigated the influence of doping reducible (Ce4+ and Cu2+) or acidic (In3+ and W6+) oxides into the SnO2 matrix. The results have illustrated that all the modified catalysts can form more abundant facile oxygen and acidic sites favorable for NH3-SCR, due to the generation of solid solutions. The samples doped by Ce4+ or Cu2+ have a good balance between reactive oxygen and acidic sites, thus displaying very good catalytic performance[21].
In addition, research on using Ce and Sn oxides in CO oxidation is also quite extensive. Liu et al. prepared a CuO/CeO2 catalyst adopting an OH-rich Ce(OH)x support, which exhibited excellent low-temperature CO oxidation activity. The reason is attributed to the enhancement of the dispersion of CuOx on CeO2 by the abundant OH species, thus increasing the CuOx-CeO2 interface and surface vacancies and promoting oxygen activation and migration[22]. Sun et al. employed SnO2 to modify CuMnOx for low-temperature CO oxidation, finding that a suitable quantity of SnO2 can improve the formation of more amorphous oxides, increase active oxygen content, and favor the dispersing of Cu and Mn. Among them, the CuMnSn8 catalyst achieved full CO conversion below 55 °C and 85% humidity[23]. The Boronin team prepared Pd/CeO2-SnO2 catalysts with different Ce/Sn ratios and found that when the ratio was below 3/1, these catalysts maintained high thermal stability and excellent low-temperature CO oxidation activity even after calcination up to 1,000 °C[24]. To further investigate the modifying effect of tin ions on the fluorite crystal structure of cerium dioxide, they synthesized complex, highly defective composite oxide catalysts, PtCeC and PtCeSnC, stabilized within a carbon matrix. Through experimental and density functional theory (DFT) calculations, it was found that the modification of the CeO2 fluorite lattice by tin induces a large number of mobile active oxygen species, thereby significantly enhancing the low-temperature CO oxidation activity[25].
Although some researchers have studied the simple CeO2-SnO2 mixed oxides and their solid solutions for catalytic NOx-SCR and CO oxidation, Ce2Sn2O7 pyrochlore-based catalysts have not yet been investigated[26]. An A2B2O7 pyrochlore compound usually possesses good thermal stability and intrinsic oxygen vacancies, which can facilitate the creation of plentiful active surface oxygen and acidic sites[27-30]. As mentioned above, these two types of reactive sites are crucial to getting high-efficiency NH3-SCR and CO oxidation catalysts. Therefore, it is worthwhile to study further the catalysis chemistry of A2B2O7 pyrochlores in NH3-SCR reaction and CO oxidation.
FeOx-based catalysts show excellent N2 selectivity and anti-deactivation to sulfur in medium-temperature for NOx-SCR reactions. Compared with single Fe2O3, the low temperature catalytic performance can be promoted by introducing secondary metal oxides, due to synergistic interaction between FeOx and other components[31,32]. For example, Han et al. found that a Fe2O3-CeO2@Al2O3 displayed over 90% NO conversion and 98% N2 selectivity in 250-430 °C, and with superior SO2 resistance. The strong Fe2O3-CeO2 interaction produces more oxygen defects and reactive oxygen sites to accelerate the redox cycle. SO2 will preferentially sulfate CeO2 to prevent the deactivation of Fe2O3, thus keeping the high reactivity of NH4+ and NH2 species[33].
The performance of A2B2O7 catalysts is closely related to their sub-crystalline phase structure, which is determined by the ratio of the elements A and B (rA/rB). At the theoretical boundary value of rA/rB, A2B2O7 paramorphs, with identical elemental compositions but exhibiting two crystalline phases, can be fabricated by regulating the temperature, pressure, or other factors[34,35]. In this work, ‘paramorphism effect’ refers to the phenomenon that two materials have identical chemical compositions (Ce/Sn = 1:1) but different crystal topologies, such as ordered pyrochlore Ce2Sn2O7 vs. biphasic Ce2Sn2O8+x solid solution compounds in this study, which exhibit significantly varied catalytic performance. This concept is used as a direct comparison of structure–activity relationships, as well as a structural regulation strategy to design efficient catalysts.
Targeting to obtain high-performance NH3-SCR and CO oxidation catalysts, Ce2Sn2O7 pyrochlore and Ce2Sn2O8+x solid solution, two paramorphs supports, were thus intentionally prepared by a hydrothermal method. 5% FeOx was loaded onto them to prepare catalysts for elimination of NO and CO together. It was revealed that the Fe/Ce2Sn2O7 pyrochlore catalyst displays better reaction performance than the Fe/Ce2Sn2O8+x solid solution catalyst. By using multiple characterization techniques and DFT calculations, the paramorphism effect of the Ce-Sn complex oxides, and their interactions with FeOx, were investigated. In addition, the reactive intermediate differences on the catalysts were explored with in-situ diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS).
2. Experimental Section
2.1 Catalyst preparation
2.1.1 Preparation of the supports
The Ce2Sn2O7 pyrochlore and Ce2Sn2O8+x solid solution supports as well as individual CeO2 and SnO2 were acquired by a hydrothermal method. For Ce2Sn2O7 and Ce2Sn2O8+x preparation, Ce(NO3)3·6H2O (AR) was used as the Ce source, while SnCl4·5H2O (AR) and Na2SnO3·3H2O (AR) were used as Sn sources, respectively, with the Ce/Sn molar ratio controlled at 1:1. With the synthesis of Ce2Sn2O7 as an example here, equal moles of Ce(NO3)3·6H2O and Na2SnO3·3H2O were dissolved in 50 mL deionized (DDI) water under magnetic stirring separately, and then mixed together. After stirring for 10 minutes, 5 mol/L NaOH solution was dripped into the mixed solution with continuous stirring until the pH was around 12. Subsequently, the mixture was stirred for additional 4 h at room temperature, which was then transferred into a Teflon-sealed autoclave of 150 mL capacity and aged at 180 °C for 24 h. Afterwards, the precipitate was vacuum filtered and washed with DDI water for several times until the filtrate was Na+ free, with total dissolved solids less than 20 ppm. The powder was dried at 110 °C for 8 h, and then calcined at 550 °C for 4 h in air with a ramp of 2 °C min-1. For the preparation of the Ce2Sn2O8+x solid solution support, the same procedure was adopted except that SnCl4·5H2O was used as the Sn precursor. For comparison purposes, single CeO2 and SnO2 were also synthesized following the same method by using Ce(NO3)3·6H2O and SnCl4·5H2O as the precursors.
2.1.2 Preparation of the supported FeOx catalysts
The supported FeOx catalysts were manufactured by a wet impregnation method. The required amount of Fe(NO3)3·9H2O (AR) was dissolved first in 20 mL DDI water and followed by vigorous stirring for 10 min. After this, a certain mass of Ce2Sn2O7 and Ce2Sn2O8+x supports as well as CeO2 and SnO2 were added into the solution to keep a FeOx mass fraction of 5 wt%. The mixtures were magnetically stirred at room temperature for 4 h to make Fe species sufficiently disperse on the surface of the supports. Subsequently, they were moved to an 80 °C water bath with continuous stirring to evaporate the excess water. After drying further in an oven at 110 °C overnight in air, the samples were grinded and calcined in a muffle room at 550 °C in air for 4 h to obtain the final catalysts, which were named Fe/Ce2Sn2O7, Fe/Ce2Sn2O8+x, Fe/CeO2, and Fe/SnO2. The elemental composition of each catalyst was confirmed with ICP analysis (Table 1).
| Catalysts | SBET (m2/g) | Lattice parameters a | FeOx content (wt. %) b | Sn/Ce mole ratio b | |||
| a (Å) | b (Å) | C (Å) | α/β/γ(°) | ||||
| Ce2Sn2O7 | 67.0 | 10.547 | 10.547 | 10.547 | 90 | 1.08 | |
| Fe/Ce2Sn2O7 | 47.6 | 10.537 | 10.537 | 10.537 | 90 | 4.7 | 1.09 |
| Ce2Sn2O8+x | 63.2 | 5.572 | 5.572 | 5.572 | 90 | 1.09 | |
| Fe/Ce2Sn2O8+x | 52.5 | 5.568 | 5.568 | 5.568 | 90 | 4.8 | 1.11 |
| CeO2 | 34.4 | 5.622 | 5.622 | 5.622 | 90 | ||
| Fe/CeO2 | 28.3 | 5.621 | 5.621 | 5.621 | 90 | 4.8 | |
| SnO2 | 22.0 | 4.741 | 4.741 | 3.190 | 90 | ||
| Fe/SnO2 | 17.6 | 4.736 | 4.736 | 3.162 | 90 | 4.7 | |
a: Calculated by Scherrer equation from XRD patterns; b: ICP-OES analysis; XRD: X-ray diffraction; ICP-OES: inductively coupled plasma-optical emission spectrometry.
2.2 Catalyst characterization and activity tests
The detailed information including the used instrument types and models, the test parameters, the experimental processes and DFT calculations are presented in the Supplementary material file.
3. Results and Discussion
3.1 Structure properties
3.1.1 X-ray diffraction (XRD) and Raman analysis
XRD patterns were collected to analyze the crystal structures. As demonstrated in Figure 1A, the typical diffraction peaks assigned to CeO2 with a cubic fluorite phase, and SnO2 with a tetragonal rutile phase are observed for individual CeO2 and SnO2, respectively[19,36]. It is mentioned that the Ce2Sn2O8+x support shows three diffraction peaks at 28.66° (111), 47.82° (220), 56.84° (311) related to cubic CeO2, and two strong peaks at 26.24° (110), 51.30° (211) attributed to rutile SnO2, indicating the coexistence of a CeO2-based cubic solid solution where Sn4+ substitutes part of the Ce4+ in the CeO2 matrix, and a SnO2-based tetragonal solid solution where Ce4+ substitutes part of the Sn4+ in the SnO2 matrix. This suggests that Ce2Sn2O8+x is a biphasic solid solution system. Interestingly, the peak positions attributed to CeO2 of the Ce2Sn2O8+x support are distinctly higher than that of pure CeO2 (28.40°, 47.32°, 56.26°), testifying that the Sn4+ with a smaller ionic radius (0.069 nm with a coordination number of 6) could be embedded into the lattice of CeO2 to form mainly a cubic CeO2-based solid solution, thus leading to unit lattice contraction[17,19]. However, a small portion of Ce4+ with a larger ionic radius (0.087 nm) could also be doped into the matrix of SnO2 to generate a tetragonal SnO2-based solid solution, thus resulting in lattice expansion. This is well supported by the moving of the 2q peaks to a lower angle (51.30°) in comparison with pure SnO2[18,21]. For the Ce2Sn2O7 support, the peaks at 29.10°, 33.64°, 48.70° and 57.84° are respectively assigned to the (222), (400), (440) and (622) planes of Ce2Sn2O7 pyrochlore (PDF#48-0640) with a space group of Fd-3m[37-39]. The lattice parameters in Table 1 have also testified that a pyrochlore phase has been formed in Ce2Sn2O7. After loading FeOx, Figure 1B displays no typical diffraction peaks assigned to FeOx, but the initial diffraction peaks of the supports become lower, which implies that FeOx is highly dispersed on the supports in an amorphous state or small clusters.
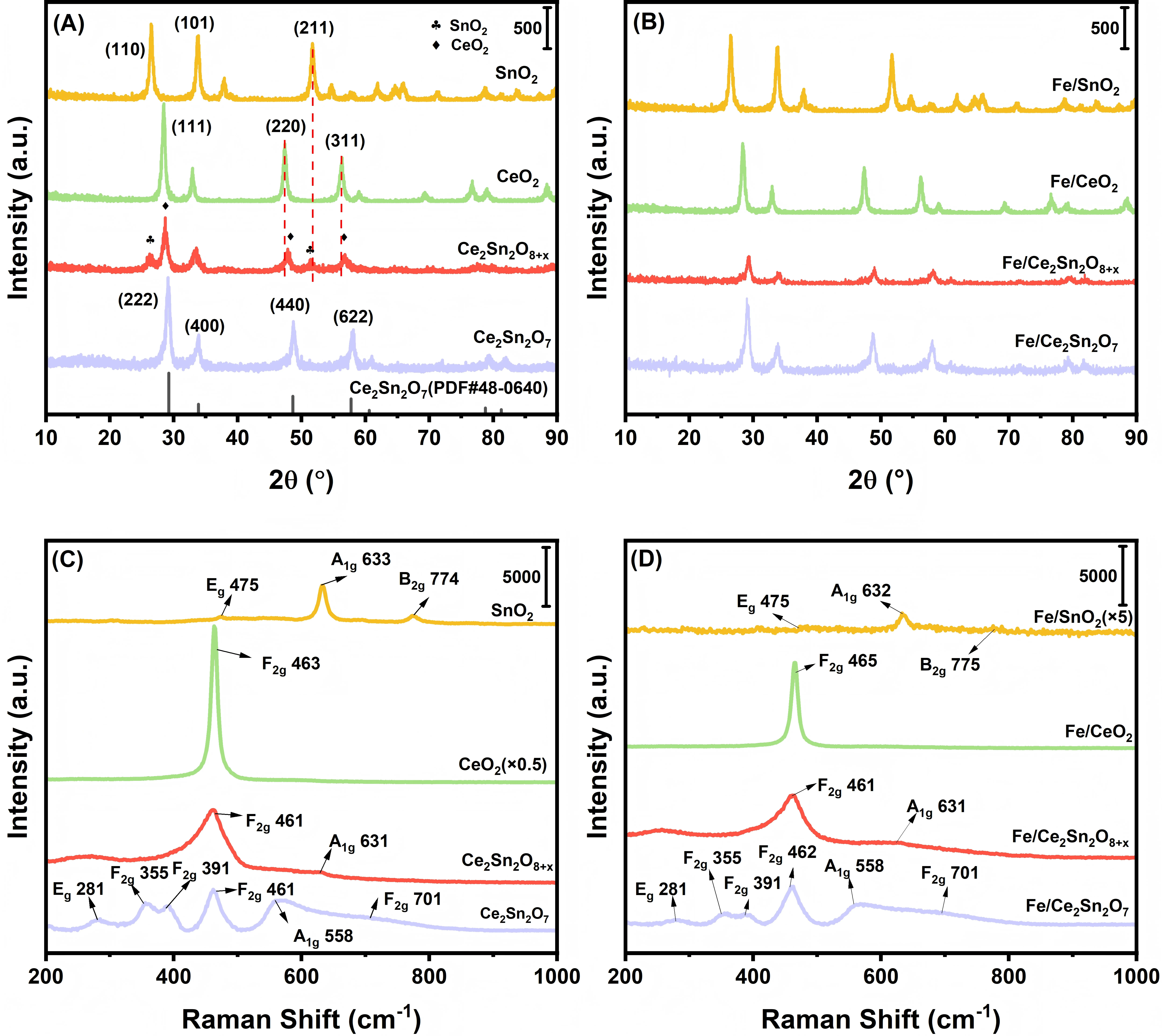
Figure 1. XRD results of the supports (A) and the catalysts (B); Raman spectra of the supports (C) and the catalysts (D). XRD: X-ray diffraction.
Raman spectra are exhibited in Figure 1C for the catalysts. Pure SnO2 shows three bands at 475, 633 and 774 cm-1, which are attributable to the Eg, A1g and B2g vibrations of the tetragonal rutile SnO2 phase[21]. Pure CeO2 exhibits a distinct peak at 463 cm-1 ascribed to the characteristic F2g mode of the symmetric vibration of Ce-O bonds[19]. In contrast, two distinctive Raman peaks are observed for the Ce2Sn2O8+x support at 461 and 631 cm-1, which are respectively assigned to the F2g mode of the CeO2 fluorite phase and the A1g mode of rutile SnO2. Compared with pure CeO2 and SnO2, the two peaks reveal shifting to lower wavenumbers and broadening, testifying again to the doping of Sn4+ into the CeO2 lattice and the doping of Ce4+ into the SnO2 lattice, which could create more lattice disorder and defects[40]. Whereas, the Raman spectroscopy of the Ce2Sn2O7 support detects 6 bands at 281, 355, 391, 461, 558, and 701 cm-1, which are typical for a pyrochlore structure[35,38,41-43]. Specifically, the largest band at 461 cm-1 is attributed to the F2g mode of Ce-O stretching vibration. Other three F2g modes at 355, 391 and 701 cm-1 are assigned to Ce-O/Sn-O stretching and O-Sn-O bending vibration in sequence. The peak at 281 cm-1 belongs to the Sn-O vibration of the Eg mode, and the peak at 558 cm-1 belongs to the A1g mode of O-Sn-O bending vibration in the SnO6 octahedra. All these characteristic Raman bands have strongly proven that a disordered pyrochlore phase is formed in Ce2Sn2O7. Figure 1D shows the Raman spectra of the FeOx catalysts. Compared with the corresponding supports, except that the intensity of all the peaks is weakened, no peak related to FeOx can be detected, confirming that the FeOx is highly dispersed, well consistent with the XRD measurements.
The specific surface areas of Ce2Sn2O7 and Ce2Sn2O8+x supports are close to each other, which are much higher than those of the individual oxides. After loading FeOx and being subjected to the second time calcination, all the surface areas are slightly decreased in comparison with the supports (Table 1).
3.1.2 Transmission electron microscopy (TEM) and high-resolution transmission electron microscopy (HRTEM) analysis
TEM, HRTEM and energy dispersive spectroscopy (EDS)-mapping were employed to analyze the samples (Figure 2). The TEM pictures in Figure 2A1,A2,A3,A4 depict that all the samples are composed of irregular grains with non-uniform sizes. The size distribution profiles in Figure 2B1,B2,B3,B4 indicate that after loading FeOx, the mean particle sizes of the samples become slightly smaller than the respective supports. As demonstrated in Figure 2C1,C2,C3,C4, the HRTEM images of the two Ce2Sn2O7 catalysts observe the lattice fringe with spacings of 0.187 and 0.305 nm, which can be attributed to the (440) and (222) facets of the cubic Ce2Sn2O7 pyrochlore phase[37]. In contrast, the images of the two Ce2Sn2O8+x samples observe the lattice fringes of both CeO2 and SnO2, confirming that both CeO2-based and SnO2-based solid solutions are present together, in agreement with the XRD and Raman results. Notably, no lattice fringes related to FeOx species are detected, confirming that FeOx is finely dispersed on the supports without forming detectable crystalline domains.
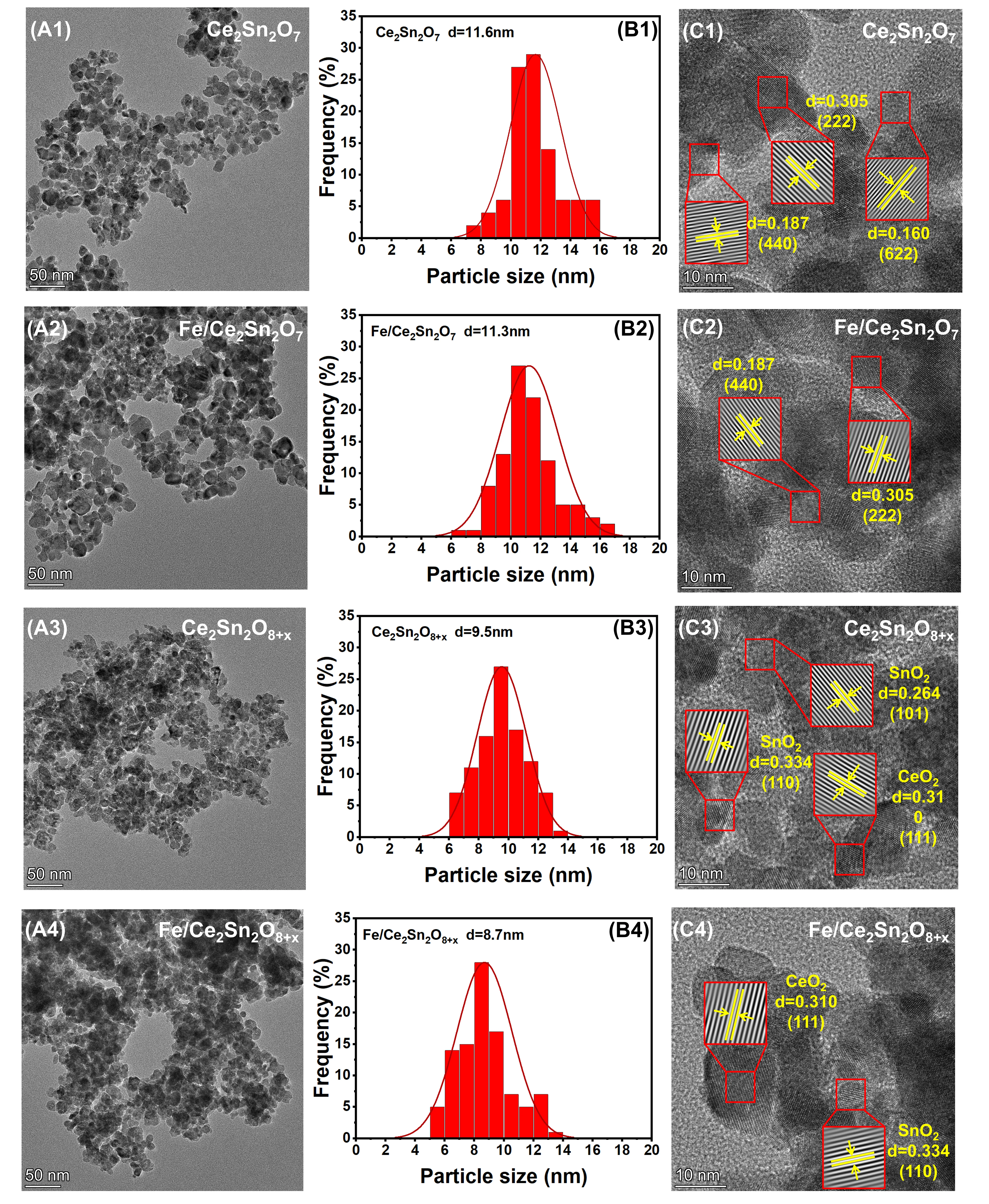
Figure 2. TEM pictures (A1-A4), particle size distribution profiles (B1-B4) and HRTEM pictures (C1-C4) and of the catalysts. TEM: Transmission electron microscopy.
As shown in Figure 3, the EDS-mapping images of Fe/Ce2Sn2O7 and Fe/Ce2Sn2O8+x catalysts demonstrate that Fe, Sn, Ce and O elements are uniformly distributed, testifying that pure phase Ce2Sn2O7 pyrochlore and Ce2Sn2O8+x solid solution are formed respectively in the catalysts. In addition, the loaded FeOx is also dispersed evenly on the supports, well consistent with the XRD, Raman and HRTEM tests.
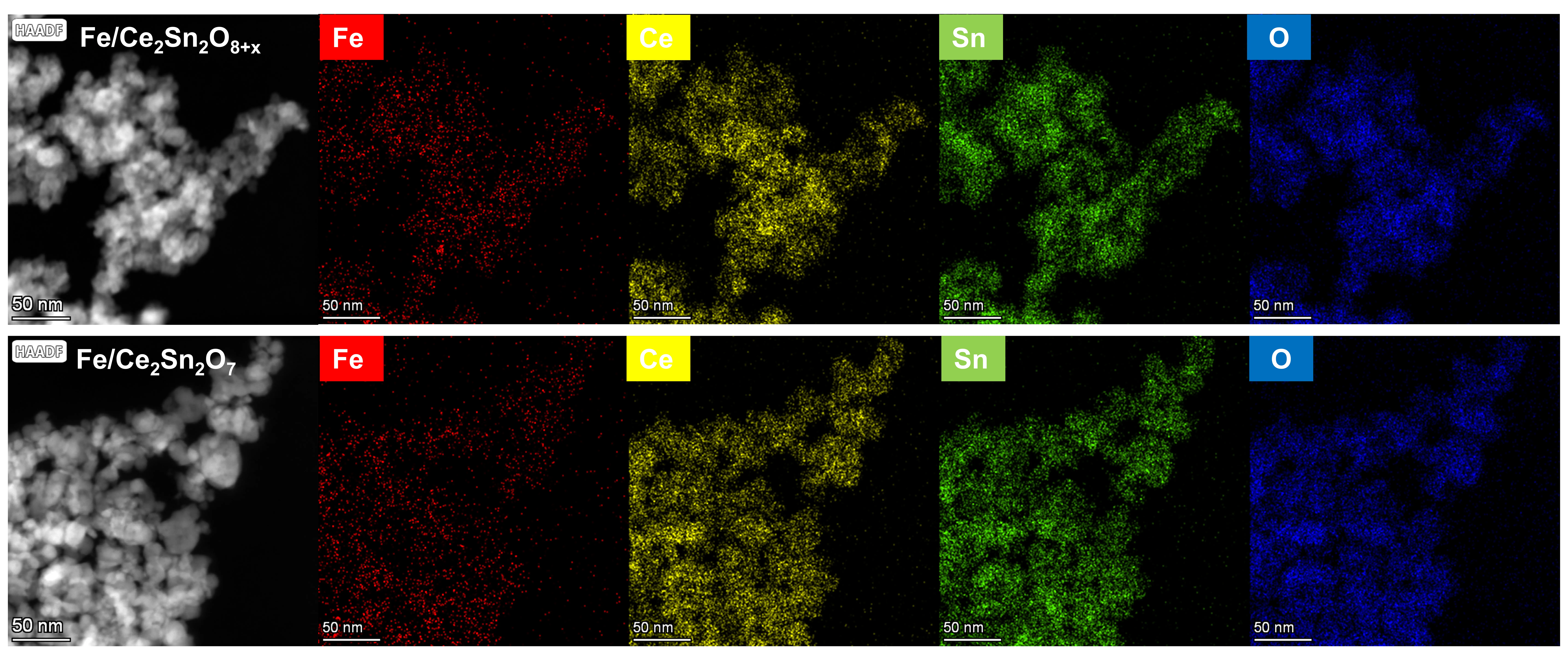
Figure 3. EDS-mapping images of Fe/Ce2Sn2O8+x and Fe/Ce2Sn2O7 catalysts. EDS: energy dispersive spectroscopy.
3.2 Catalytic performance
The performance of all catalysts was evaluated for the simultaneous removal of NO and CO. As depicted in Figure S1, individual CeO2 and SnO2 exhibit low NO and CO conversion. After loading FeOx, Fe/CeO2 and Fe/SnO2 have only mildly improved reaction performance. In contrast, the performance on the Ce2Sn2O7 pyrochlore and Ce2Sn2O8+x solid solution supports is evidently higher, achieving a maximum NO conversion above 85% around 350 °C. As manifested in Figure 4A, after supporting FeOx, the NH3-SCR performance is significantly improved in comparison with the Ce-Sn supports at the low temperature region. On the optimal Fe/Ce2Sn2O7 catalyst, a NO conversion of > 90% is maintained within 275-400 °C, while 100% CO oxidation is achieved at 200 °C. The activity for the simultaneous removal of NO and CO is significantly improved compared to that of the Ce2Sn2O7 support. For the Ce2Sn2O8+x sample, after loading FeOx, the activity of the Fe/Ce2Sn2O8+x shows a milder improvement. Figure 4B displays that in the whole tested temperature region, both Fe/Ce2Sn2O7 and Fe/Ce2Sn2O8+x, and the corresponding supports exhibit high N2 selectivity, maintaining > 90% within 100-400 °C.
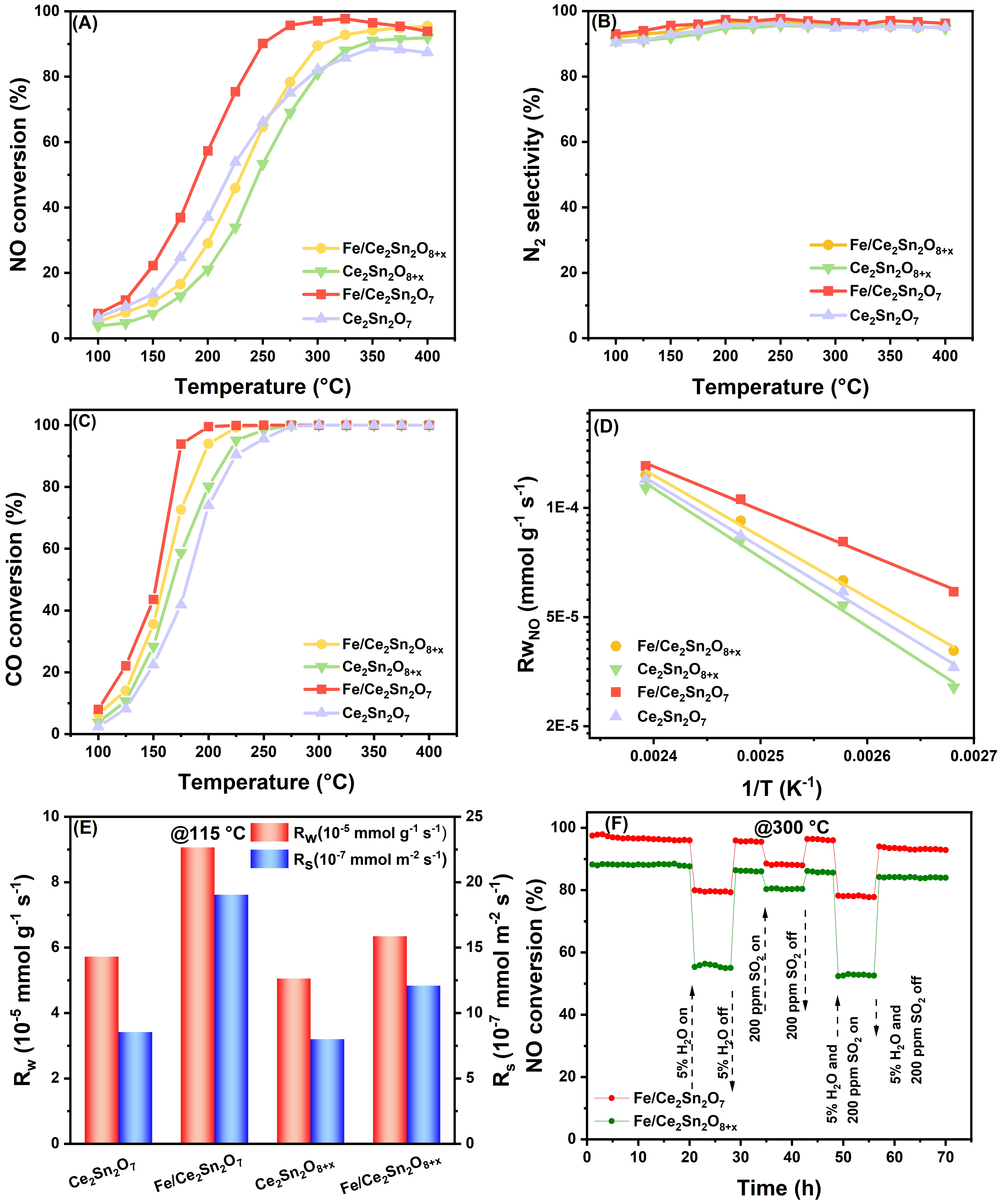
Figure 4. NH3-SCR performance tests. (A) NO conversion; (B) N2 selectivity; (C) CO conversion; (D) Arrhenius plots; (E) NO conversion rates (Rw and Rs) at 115 °C; (F) stability tests of Fe/Ce2Sn2O7 and Fe/Ce2Sn2O8+x catalysts with H2O and/or SO2 at 300 °C; (Reaction conditions: 10 mg catalyst, [NH3] = [NO] = 500 ppm, [CO] = 2,000 ppm, [O2] = 5 %, N2 balance, GHSV = 600,000 h-1). NH3-SCR: NH3-selective catalytic reduction; GHSV: gas hourly space velocity.
To elucidate the intrinsic activity change of the catalysts, the Arrhenius plots of Fe/Ce2Sn2O7, Fe/Ce2Sn2O8+x and the corresponding supports are plotted in Figure 4C. In a testing process, the NO conversion is always controlled below 10% to minimize the effect of diffusion and heat transfer. The NO conversion rates normalized by catalyst weight (Rw) and surface area (Rs) were achieved from 100 to 145 °C by adjusting the catalyst mass. The activation energies extracted from Figure 4D are listed in Table S1, which indicates that Fe/Ce2Sn2O7 has the lowest activation energy (33 kJ/mol), among all the catalysts, significantly lower than that of Fe/Ce2Sn2O8+x (46 kJ/mol). This indicates that Fe/Ce2Sn2O7 possesses the strongest ability to activate NO molecules, which is attributed to its higher density of oxygen vacancies and active oxygen species derived from the pyrochlore structure. The Rw and Rs collected at 115 °C on the catalysts are particularly compared in Figure 4E. It is apparent that with the addition of FeOx, the intrinsic activity on both Fe/Ce2Sn2O7 and Fe/Ce2Sn2O8+x are enhanced in comparison with the supports. This indicates that the dispersed FeOx interacts with the supports to create more reactive sites, thus improving the NH3-SCR performance. Figure S2 shows the RS for Fe/Ce2Sn2O7 and Fe/Ce2Sn2O8+x at 200-300 °C. It can be clearly seen that the RS of Fe/Ce2Sn2O7 are significantly higher than that of the Fe/Ce2Sn2O8+x, which strongly proves the impact of the paramorphism effect.
The reaction stability and the capability to tolerate SO2 and H2O deactivation were tested for Fe/Ce2Sn2O7 and Fe/Ce2Sn2O8+x. As displayed in Figure 4F, for both catalysts, the presence of SO2 and/or H2O imposes only recoverable deactivation, testifying that SO2 and H2O compete merely for the active sites with the reactants, but have no permanent damage to the surface structure. However, it can be seen that Fe/Ce2Sn2O7 has better water resistance than Fe/Ce2Sn2O8+x. The long-term (50-100 hours) stability tests shown in Figure S3, show that under SO2-only conditions, both catalysts show stable NO conversion over 50 h with only slight decreases. However, upon H2O introduction, Fe/Ce2Sn2O7 exhibits much less activity loss than Fe/Ce2Sn2O8+x, confirming its superior resistance to both H2O and SO2. The pyrochlore structure of Ce2Sn2O7 plays a key role in sulfur and water vapor poisoning. First, its higher oxygen vacancy density promotes SO2 oxidation to surface sulfates, sacrificially protecting FeOx sites. Second, the flexible pyrochlore lattice and Ce3+/Ce4+ redox cycles allow reversible sulfate decomposition, explaining the recoverable deactivation. Third, the exclusive Brønsted acid sites (NH4+) are less competitive with H2O than Lewis acid sites, enhancing its water resistance. Considering all these issues, it is concluded that Fe/Ce2Sn2O7 is the optimal catalyst in this work, which shows promising application potential.
3.3 H2-TPR and O2-TPD analysis
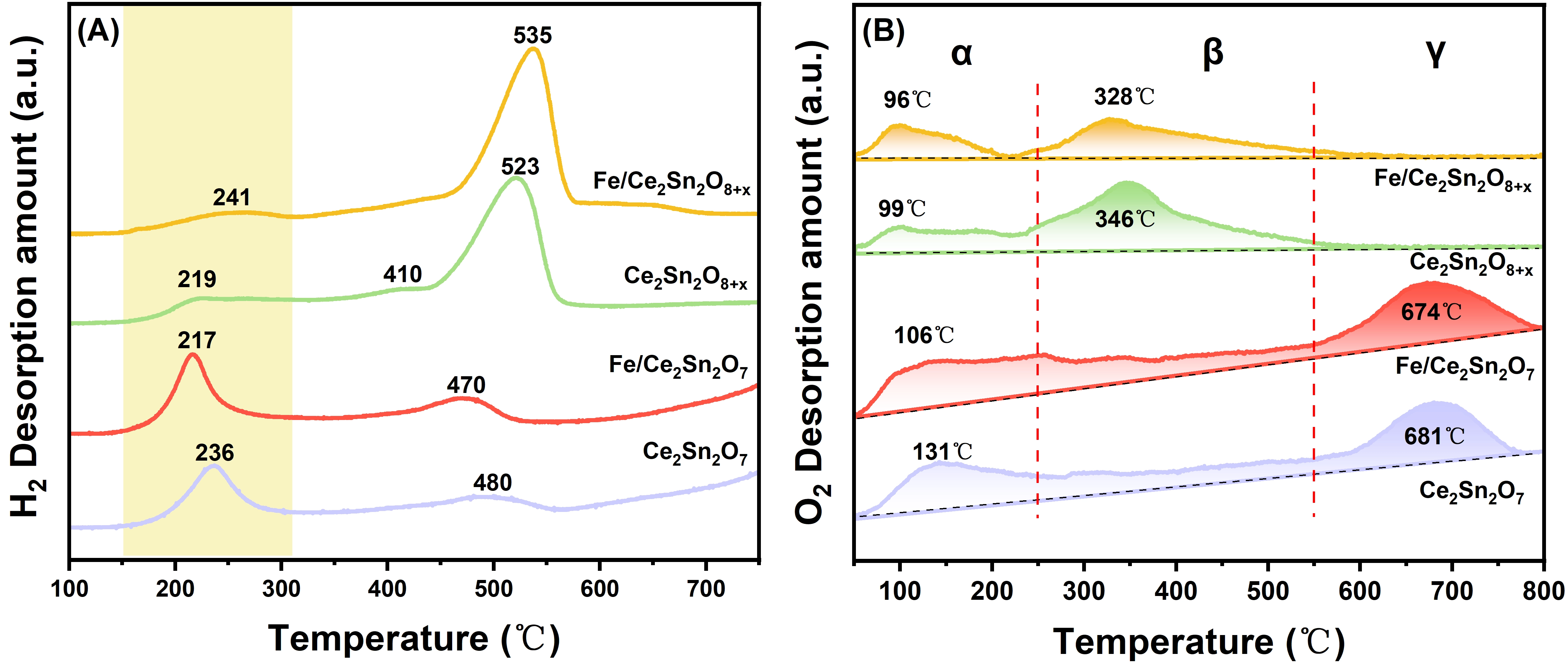
The redox and oxygen property is usually a key factor to determine the NH3-SCR performance[10]. Thus, H2-TPR and O2-TPD were adopted to analyze the catalysts as well as the supports. Figure 5A exhibits the H2-TPR profiles and Table S2 presents the respective quantitative results. For all the samples, there are mainly two reduction peaks present in 150-300 °C and 450-600 °C. The first peak is assigned to the reduction of chemisorbed oxygen, and the second peak is ascribed to the reduction of Sn4+ to metallic Sn0[17,28,44]. Notably, the H2 consumption of the low-temperature peaks of the two Ce2Sn2O7 samples is clearly higher than that of the two Ce2Sn2O8+x samples (Table S2), demonstrating that more chemisorbed oxygen species promoting the NH3-SCR performance at lower temperature are formed on the pyrochlore samples. After loading FeOx onto the Ce2Sn2O7 support, the H2 consumption of the low temperature peak is further improved, and the peak position moves to a lower temperature. This illustrates that the interaction of FeOx and the Ce2Sn2O7 support can promote the generation of more active oxygen sites. However, loading FeOx onto the Ce2Sn2O8+x solid solution support can suppress slightly the chemisorbed oxygen amount. As a result, Fe/Ce2Sn2O7 owns a richer amount of chemisorbed oxygen species than Fe/Ce2Sn2O8+x, thereby showing better catalytic performance.
The O2-TPD curves are displayed in Figure 5B, which can be classified into three types. The α peak below 250 °C is related to the desorption of loosely chemisorbed oxygen species. The β peak between 250 and 550 °C is ascribed to the release of chemisorbed surface oxygen species with medium strength. And the γ peak over 550 °C might be assigned to the release of surface lattice oxygen, which could have limited contribution to the reaction[45]. As quantified in Table S3, the desorbed oxygen amount of Fe/Ce2Sn2O7 corresponding to α + β peaks is higher than that over Fe/Ce2Sn2O8+x, being well consistent with the H2-TPR results. Indeed, the H2-TPR and O2-TPD results have demonstrated that the number of surface-active oxygen sites might be a vital factor to influence the catalytic performance over the two supported FeOx catalysts.
3.4 EPR and DFT calculation study on the surface vacancies
EPR was employed to certify the existence of oxygen vacancies and evaluate their concentration on Fe/Ce2Sn2O7 and Fe/Ce2Sn2O8+x catalysts. The EPR spectra in Figure 6A show a signal at g = 2.003 for both catalysts, which can be assigned to surface oxygen vacancies[2,46]. Furthermore, the signal intensity of Fe/Ce2Sn2O7 is distinctly higher than that on Fe/Ce2Sn2O8+x, indicating the formation of more oxygen vacancies on Fe/Ce2Sn2O7. It is well agreed that the surface oxygen vacancies can facilitate the chemisorption of gas phase O2 to create surface-active oxygen species[47-49]. A catalyst with a richer amount of oxygen defects usually possesses more surface active oxygen species. This is in good agreement with the H2-TPR and O2-TPD findings that Fe/Ce2Sn2O7 owns more abundant active oxygen sites than Fe/Ce2Sn2O8+x, thus showing better NH3-SCR and CO oxidation performance.
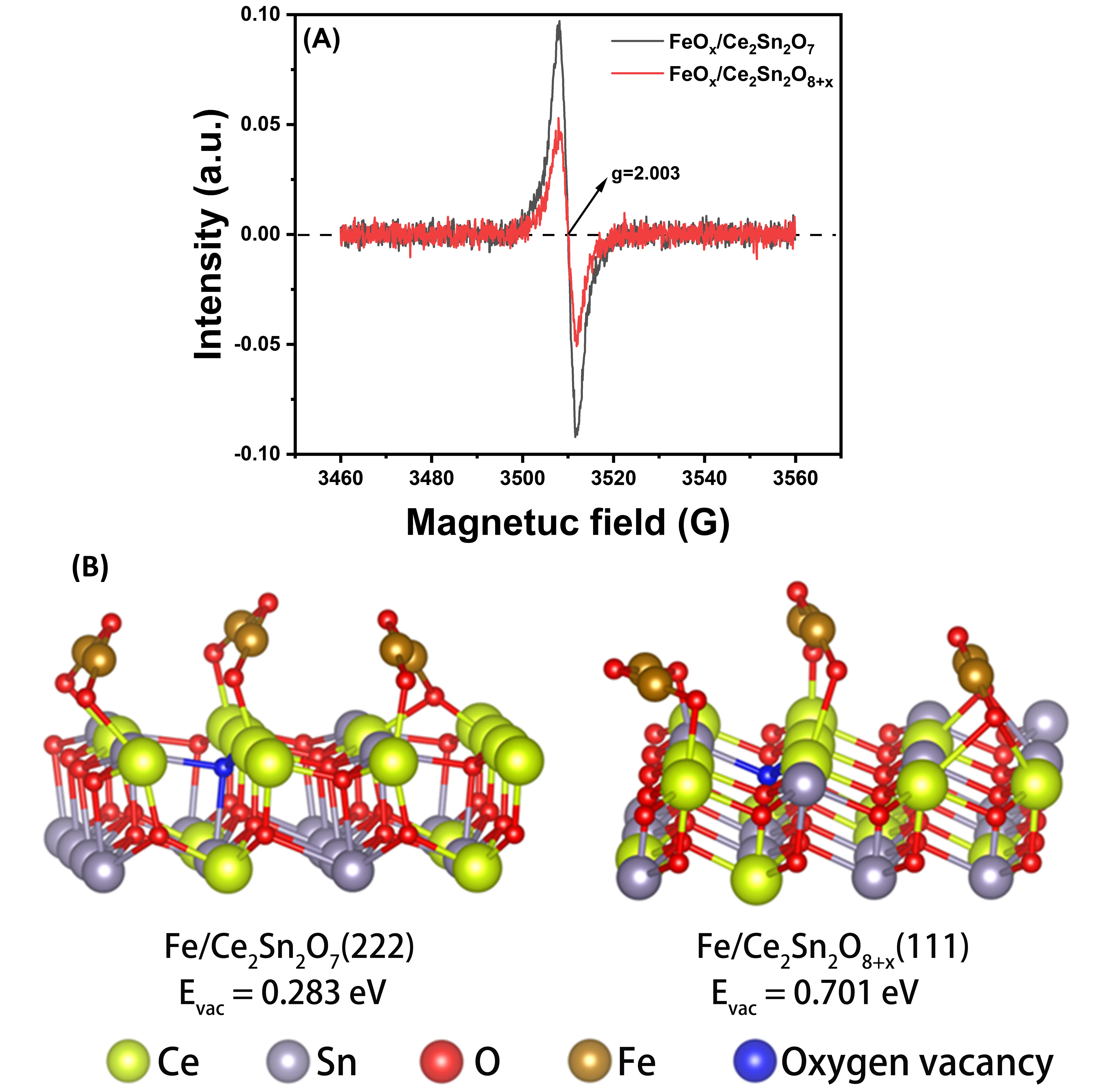
Figure 6. (A) EPR spectra and (B) DFT calculation results and models for oxygen vacancy formation energies of Fe/Ce2Sn2O7 and Fe/Ce2Sn2O8+x catalysts. EPR: electron paramagnetic resonance; DFT: density functional theory.
To further understand the surface defects property on the two paramorphic catalysts, a DFT method was adopted to calculate the oxygen vacancy formation energies of Fe/Ce2Sn2O7 and Fe/Ce2Sn2O8+x. Considering the XRD and HRTEM results, the (222) planes on the Ce2Sn2O7 pyrochlore and the (111) planes on the Ce2Sn2O8+x solid solution are employed to construct DFT models, as shown in Figure 6B. The calculations are performed via the Vienna Ab initio Simulation Package (VASP) code. Apparently, the formation energy of oxygen vacancies on the Fe/Ce2Sn2O7 catalyst is lower than that on Fe/Ce2Sn2O8+x, illustrating that it is easier to form oxygen vacancies on the Fe/Ce2Sn2O7 catalyst. This is in good accordance with the EPR, H2-TPR and O2-TPD results, validating that the surface oxygen defects and active oxygen species are important for the reaction, which could be one of the key factors in deciding the catalytic performance.
3.5 Surface acid property
The adsorbing and activating of NH3 on the acid sites is a crucial step in NH3-SCR reaction. Thus, NH3-TPD was performed for the catalysts and the respective supports. As shown in Figure S4, the main NH3 desorption peak is detected below 300 °C for all the samples, which can be ascribed to the release of NH3 adsorbed on weak acid sites[50,51]. Notably, the Fe/Ce2Sn2O7 and Fe/Ce2Sn2O8+x catalysts show similar NH3 desorption behavior to the corresponding supports, but the temperature shifts to a lower region. Moreover, the addition of FeOx improves also the NH3 desorption amounts for both samples (Table 2). Interestingly, Fe/Ce2Sn2O7 possesses obviously more surface acidic sites than the Fe/Ce2Sn2O8+x catalyst. Taking into account the reaction performance, the abundance of surface acidity could be another critical element to decide the reaction performance.
| Catalysis | NH3 Desorption | |
| T (°C) | Total acidic sites amount (mmol g-1) | |
| Ce2Sn2O7 | 133 | 0.26 |
| Fe/Ce2Sn2O7 | 99 | 0.51 |
| Ce2Sn2O8+x | 112 | 0.30 |
| Fe/Ce2Sn2O8+x | 101 | 0.37 |
To elucidate further the NH3 adsorption behavior, in-situ DRIFTS spectra of NH3 adsorbing at 50 °C on the catalysts and corresponding supports were collected in Figure 7. After the introduction of 1,000 ppm NH3/Ar for 30 min, multiple peaks belonging to NH3 adsorption are detected. The peaks at 1,158, 1,201, 1,287 and 1,559 cm-1 are assignable to NH3 attached on Lewis acidic sites (L-NH3). And the peaks at 3,389 and 3,243 cm-1 are respectively assigned to the stretching vibration of N-H[17,52]. Interestingly, two special bands at 1,442 and 1,680 cm-1, which are respectively assigned to NH4+ species attached to Brønsted acidic sites (B-NH4+)[53,54], are only detected on the two Ce2Sn2O7 samples. These B-NH4+ peaks are obviously not observed on the two Ce2Sn2O8+x samples. The ordered A2B2O7 pyrochlore lattice contains intrinsic O′ sites that are easily hydroxylated to form M–OH[55]. The strong Lewis acidity of Sn4+ polarizes adjacent O–H bonds, enhancing their proton-donating ability to NH3 (forming NH4+). Additionally, the high density of oxygen vacancies (EPR/DFT) provides unsaturated metal sites that facilitate H2O dissociation, generating more surface hydroxyls, some of which act as Brønsted acids. The biphasic solid solution lacks these structural features, explaining the absence of NH4+ in DRIFTS. It was reported that the presence of Brønsted acidic sites is favorable to enhance NH3-SCR performance at medium and low temperature[56]. Indeed, this could be an important reason that Fe/Ce2Sn2O7 exhibits better reaction performance than Fe/Ce2Sn2O8+x. It is mentioned here that loading FeOx onto the supports can increase markedly the quantities of both Lewis and Brønsted acidic sites, which are beneficial to the reaction performance.
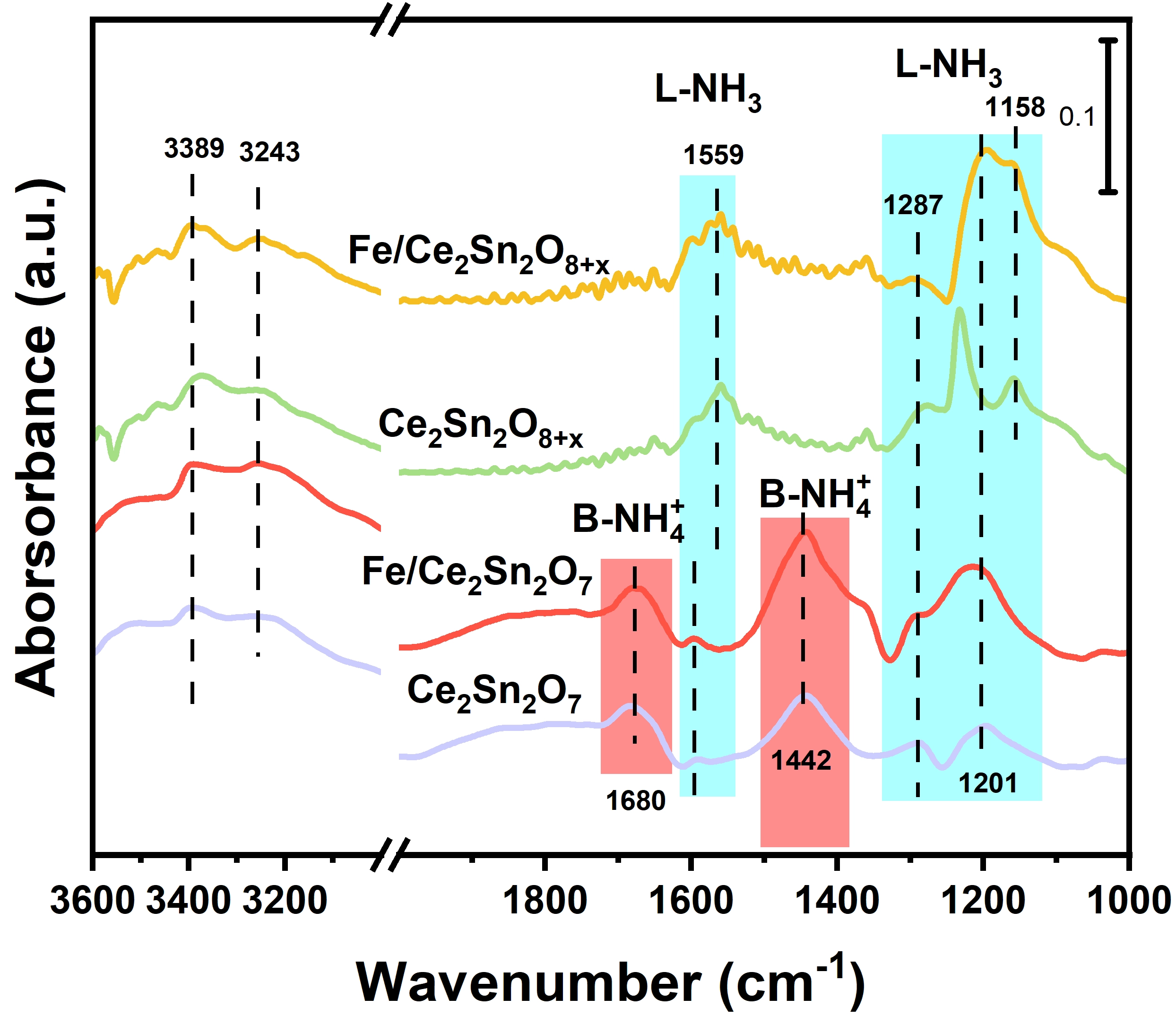
Figure 7. In-situ DRIFTS test of NH3 adsorption at 50 °C for 30 min on Ce2Sn2O8+x, Ce2Sn2O7, Fe/ Ce2Sn2O8+x and Fe/Ce2Sn2O7 catalysts. DRIFTS: diffuse reflectance infrared Fourier transform spectroscopy.
3.6 NOx storage property
Given that the activation of NOx molecules on a catalyst is vital during the NH3-SCR process, the NOx storage capacity was evaluated by performing NO + O2-TPD experiments. Notably, the effluent was monitored by a gas analyzer, thus the NO and NO2 curves in Figure 8 reflect the real NO and NO2 concentrations. All the samples show two desorption peaks in both NO and NO2 profiles. The peak below 200 °C is assigned to the release of weakly adsorbed NOx and unstable nitrates; and the peak above 200 °C is ascribed to the release of more strongly attached NOx with better thermal stability[57]. In comparison with the supports, it is apparent that the NOx desorption amount increases after loading FeOx, testifying the increase of the NOx storage capacity (Table S4). Moreover, Fe/Ce2Sn2O7 catalyst exhibits lower desorption temperature than the support. Particularly, the high temperature peak moves significantly from 300 to 209 °C. This indicates that besides the capacity improvement, the adsorbed NOx becomes also more facile. Whereas, for Fe/Ce2Sn2O8+x catalyst, adding FeOx increases both peaks to higher temperature evidently, especially the high temperature peak. This testifies that on Fe/Ce2Sn2O8+x, though the amount of adsorbed NOx is larger than that of the support, it becomes less active. According to the literatures, the presence of uncoordinated electrons in the NO molecule makes it preferentially adsorbed on the catalyst surface to form nitrate intermediates[58]. In addition, the surface active oxygen species can not only improve the formation of surface nitrates, but also promote the breakdown of nitrates or nitrites via weakening N-O bonds[59,60]. As demonstrated above, Fe/Ce2Sn2O7 owns more abundant active oxygen sites than Fe/Ce2Sn2O8+x, thus forming more reactive adsorbed NOx species. In other words, although the total NOx capacity of Fe/Ce2Sn2O8+x is higher than that of Fe/Ce2Sn2O7, the adsorbed NOx species is more stable. As a result, Fe/Ce2Sn2O7 displays higher NH3-SCR reaction performance than Fe/Ce2Sn2O8+x.
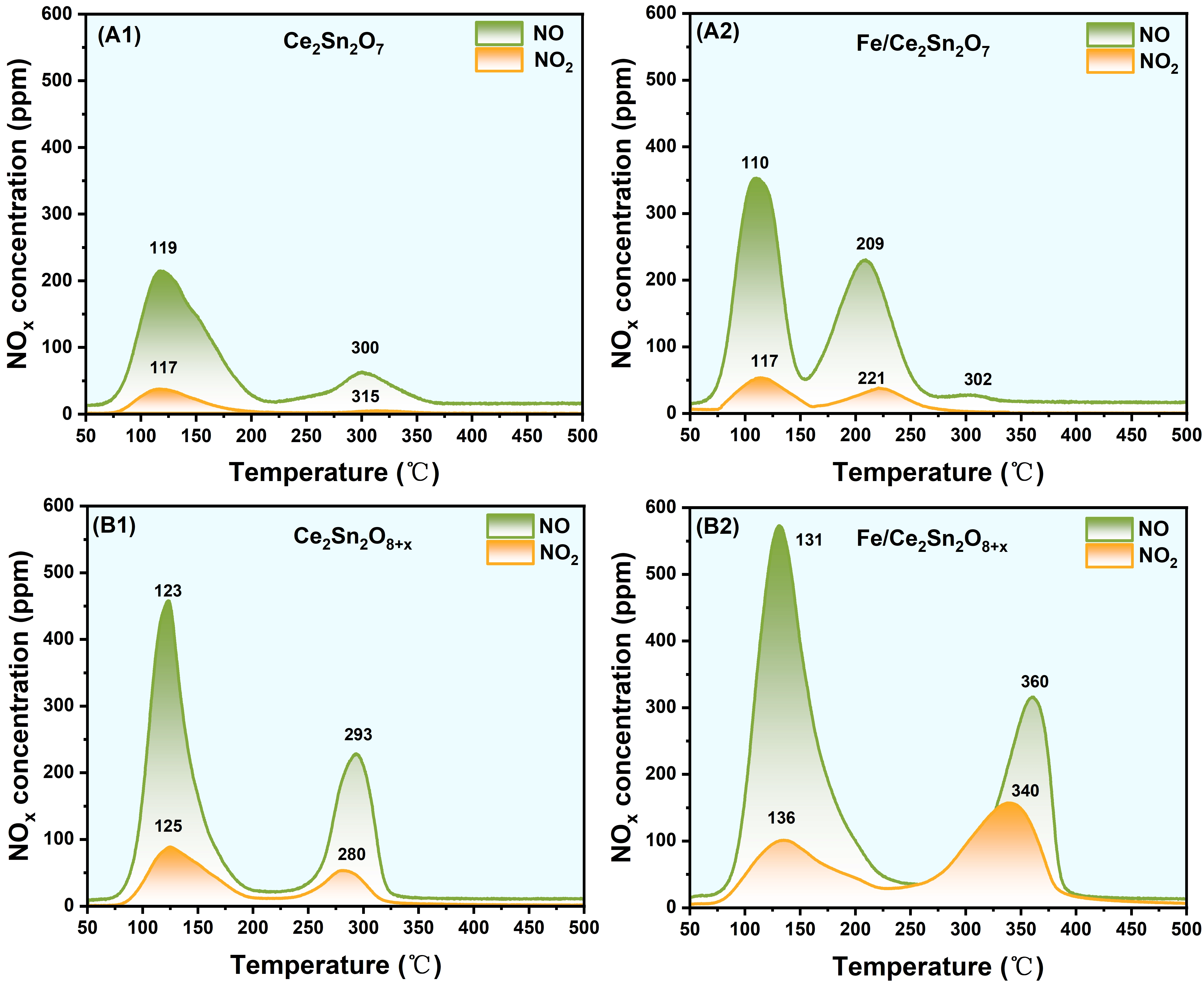
Figure 8. NO+O2-TPD profiles of Ce2Sn2O7 (A1), Fe/Ce2Sn2O7 (A2), Ce2Sn2O8+x (B1) and Fe/Ce2Sn2O8+x (B2) catalysts.
3.7 Surface chemical state analyzed by X-ray photoelectron spectroscopy (XPS)
XPS tests were performed to investigate the valence states over the catalysts and corresponding supports. The C 1s spectra of the samples are demonstrated in Figure S5. Apart from the C−C bond peak at 284.8 eV of the carbon standard, the other two peaks at 286.4 and 288.5 eV are respectively assigned to C−O and C=O bands of CO32- species in sequence[61]. This suggests that surface carbonate species are formed on the samples and contribute to the O 1s signal. Based on this, the O 1s spectra are closely analyzed for the catalysts and displayed in Figure 9A, which can be fitted into four peaks. The peaks at ~529.7, ~530.8, 532.0 and 533.0 eV are assigned to surface lattice oxygen (O2-), O22-, OH-/CO32- and O2- species in sequence[27]. As quantified in Table 3, the amount of chemical adsorbed oxygen (O22-+O2-) on Fe/Ce2Sn2O7 is obviously higher than that on Fe/Ce2Sn2O8+x, matching well with the H2-TPR and O2-TPD, EPR and DFT calculation results. In Figure 9B, the Fe 2p XPS spectra prove that on both Fe/Ce2Sn2O7 and Fe/Ce2Sn2O8+x, the Fe3+ peak at 716.1 eV and Fe2+ peak at 712.3 eV are detected, but the Fe3+ ions are denominated on the surfaces[62]. As shown in Table 3, the Fe3+ percentage/amount on both catalysts is very close, indicating that it is not the main reason to discriminate their NH3-SCR and CO oxidation performance, though the presence of FeOx sites is important for the reaction.
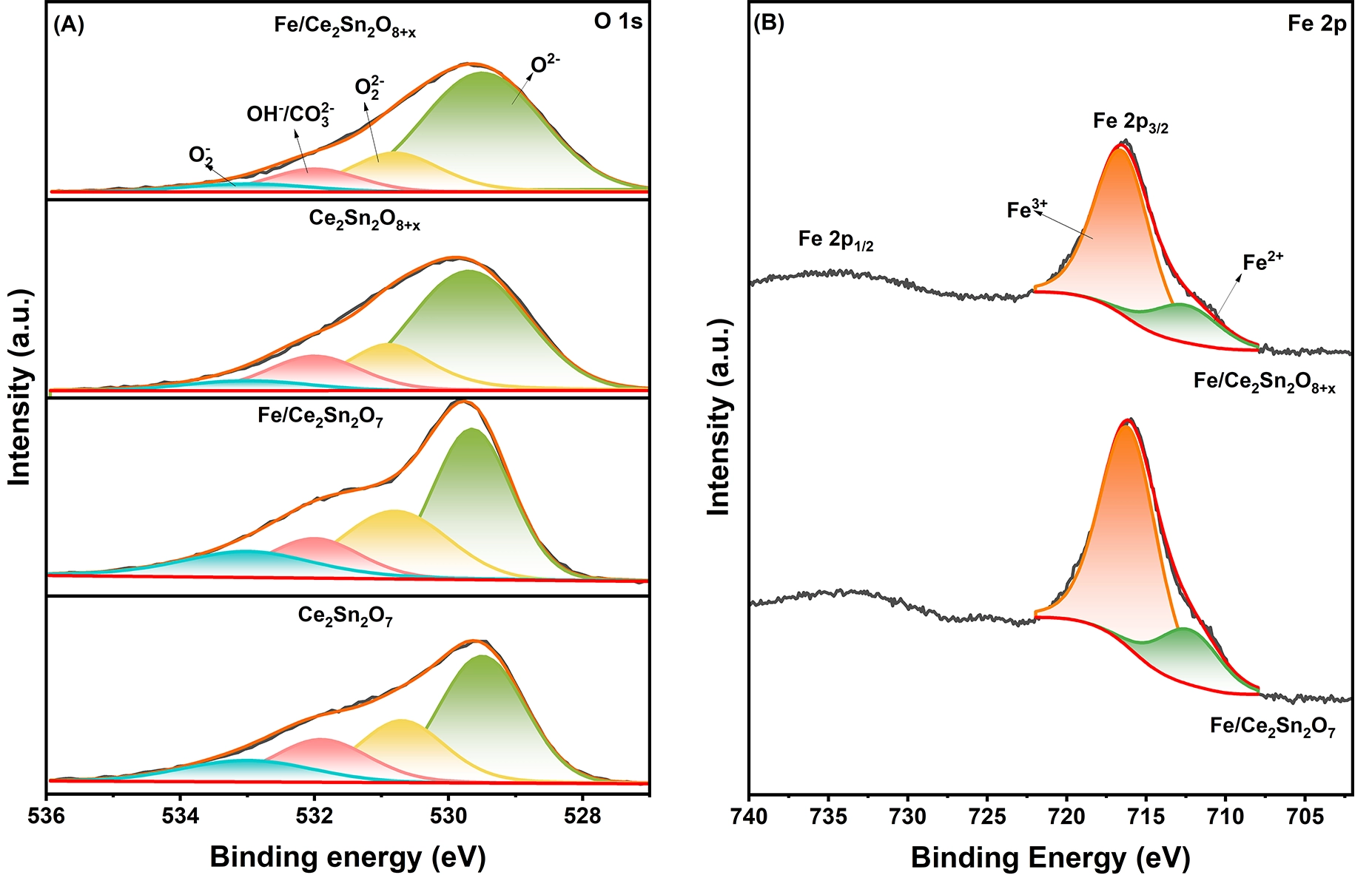
Figure 9. XPS of O 1s (A) and Fe 2p (B) on the catalysts. XPS: X-ray photoelectron spectroscopy.
| Catalysts | O 1s Binding Energy (eV) | (O22-+O2-)/Otot (%) | Ce3+/Cetot (%) | Fe3+/Fetot (%) | |||
| O2- | O22- | OH-/CO32- | O2- | ||||
| Ce2Sn2O7 | 529.6 | 530.7 | 532.0 | 533.0 | 36.5 | 41.2 | -- |
| Fe/Ce2Sn2O7 | 529.7 | 530.8 | 532.0 | 533.0 | 41.5 | 44.4 | 80.4 |
| Ce2Sn2O8+x | 529.7 | 530.9 | 532.0 | 533.0 | 26.6 | 27.2 | -- |
| Fe/Ce2Sn2O8+x | 529.6 | 530.8 | 532.0 | 533.0 | 23.4 | 26.5 | 80.3 |
XPS: X-ray photoelectron spectroscopy.
The Ce 3d XPS signals of the catalysts and respective supports are displayed in Figure S6A, which can all be fitted into eight peaks. The six bands denoted as v, v″, v‴, u, u″ and u‴ are associated with surface Ce4+, and the other two peaks marked as u′ and v′ are related to Ce3+[18]. Either forming Ce2Sn2O7 pyrochlore or Ce2Sn2O8+x solid solution, both Ce3+ and Ce4+ are present on the surface, but obviously in different amounts. The Ce3+/Cetotal percentages are calculated in Table 3 for all the samples via the fitted peak areas. It is apparent that Fe/Ce2Sn2O7 possesses much more surface Ce3+ cations than Fe/Ce2Sn2O8+x, which could be favorable to promote the chemisorption of O2 molecules to generate active surface oxygen sites[49,63].
As exhibited in Figure S6B, all the catalysts and corresponding supports have two typical Sn 3d peaks around 486.1-486.9 and 494.4-495.2 eV, which are respectively assigned to the Sn 3d5/2 and Sn 3d3/2 spin-orbit peak of Sn4+ cations. Compared with the two Ce2Sn2O8+x solid solution samples, the binding energy of Sn 3d peaks of the two Ce2Sn2O7 pyrochlore samples shifts evidently to lower values, indicating that Sn4+ cations in the matrix have higher electron density. Indeed, this could also help the chemisorption of gas phase O2, thus generating more abundant active surface oxygen anions with negative charge.
3.8 In-situ DRIFTS study on the reaction intermediates
To identify the possible intermediates and reaction pathways, three series of in-situ DRIFTS tests have been performed on both Fe/Ce2Sn2O7 and Fe/Ce2Sn2O8+x, catalysts. Before the experiments, the samples were first processed in a 30 mL/min Ar feed at 400 °C for 1 h to get rid of the surface impurities, and then cooled to 150 °C.
3.8.1 Reaction between NH3 and pre-adsorbed NO+O2 species
In this series of tests, a feed mixture of 500 ppm NO + 5vol% O2 + Ar balance was directed to the catalysts and saturated their surfaces at 150 °C. As demonstrated in Figure 10A1,B1, both Fe/Ce2Sn2O7 and Fe/Ce2Sn2O8+x catalysts exhibit similar NOx adsorption behavior. In detail, three peaks at 1,556, 1,548 and 1,300 cm-1 assigned to bidentate nitrate, and other three bands at 1,431, 1,359 and 1,197 cm-1 belonging to trans-N2O22-, cis-N2O22- and bridge nitrite are detected, respectively[5,14,64]. Notably, the peaks at 1,556 and 1,548 cm-1 increased obviously with the time extension to 40 min for both catalysts. Afterwards, all the peaks remained unchanged, indicating that the catalyst surfaces were saturated. Notably, Fe/Ce2Sn2O8+x catalyst adsorbed more NOx than Fe/Ce2Sn2O7, in agreement with the NO + O2-TPD results.
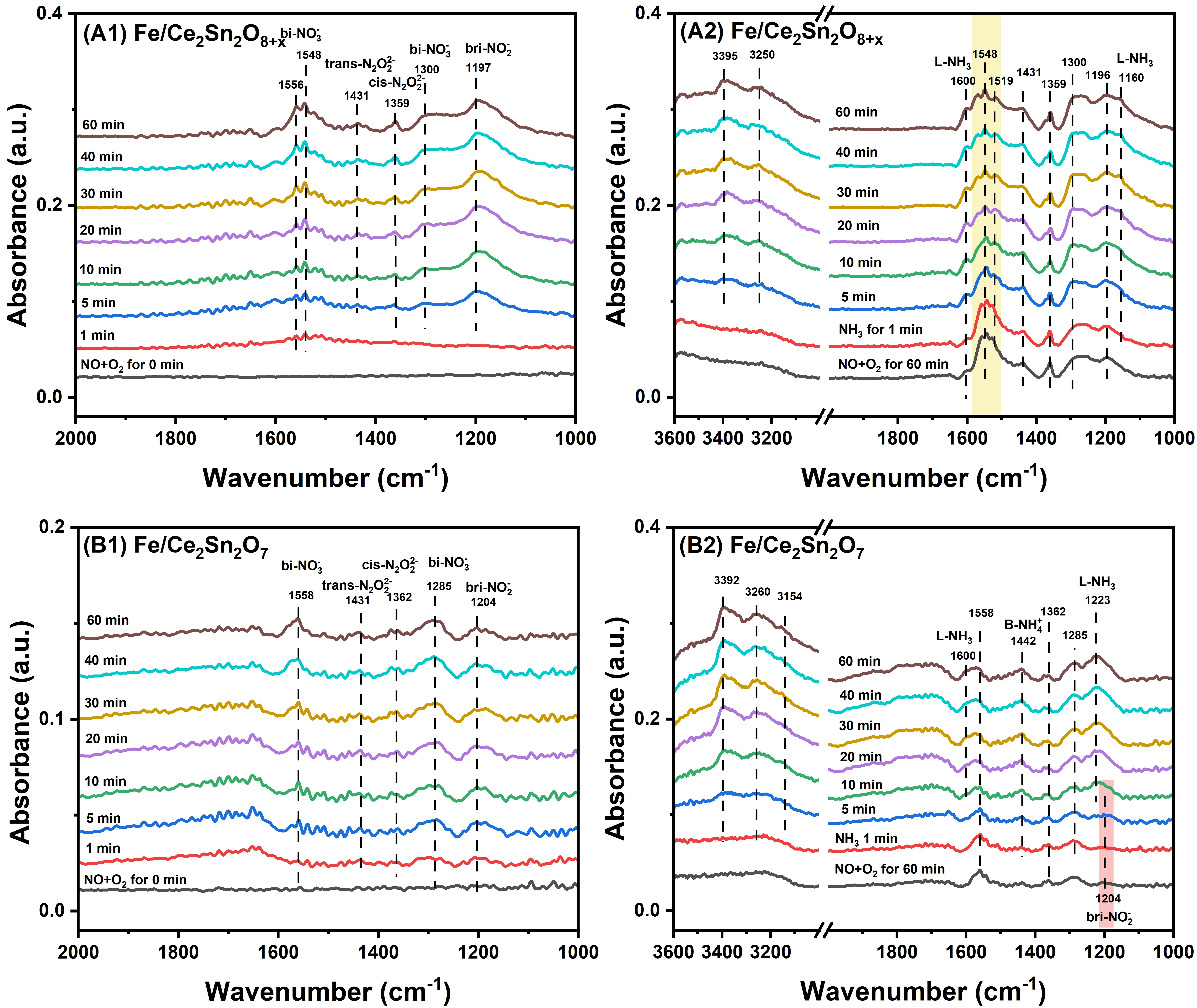
Figure 10. In-situ DRIFTS study on Fe/Ce2Sn2O8+x and Fe/Ce2Sn2O7 catalysts at 150 °C; (A1) and (B1) in a 500 ppm of NO + 5% O2/Ar flow, (A2) and (B2) in a 500 ppm of NH3/Ar flow after pre-adsorbing NO + O2. DRIFTS: diffuse reflectance infrared Fourier transform spectroscopy.
Following this step, the saturated catalysts were purged with an Ar flow to get rid of the physically adsorbed NOx species. Afterwards, a 500 ppm NH3 + Ar flow was switched to the catalyst. Figure 10A2 displays that on Fe/Ce2Sn2O8+x solid solution catalyst, the bidentate nitrate species at 1,556 and 1,548 cm-1 gradually decreased and became stable after 30 min, while the peaks corresponding to other nitrate species remained nearly unchanged. In addition, with the increase of the exposing time in NH3, new peaks at 3,395, 3,250, 1,600 and 1,160 cm-1 were observed. The bands at 3,395 and 3,250 cm-1 are related to N-H vibration of NH3, and the bands at 1,600 and 1,160 cm-1 belong to NH3 adsorbed on Lewis acidic sites[65]. This indicates that bidentate nitrates might be the active intermediates for NH3-SCR on Fe/Ce2Sn2O8+x.
In contrast, as displayed in Figure 10B2, over the Fe/Ce2Sn2O7 pyrochlore catalyst, the peak of bridge nitrite at 1,204 cm-1 decreased and disappeared in 5 minutes. At the same time, the intensity of the bidentate nitrates decreased gradually. This indicates that the bridge nitrite could be another active intermediate besides the bidentate nitrates on the Fe/Ce2Sn2O7 catalyst. Notably, a new peak at 1,442 cm-1 assigned to NH4+ species on the Brønsted acidic sites can be detected on Fe/Ce2Sn2O7, in good accordance with the NH3 adsorption in-situ DRIFTS results in Figure 7. In brief, these results testified that the bidentate nitrate could be the crucial active surface intermediates for both Fe/Ce2Sn2O7 and Fe/Ce2Sn2O8+x catalysts in NH3-SCR reaction. The additional bridge nitrite formed on Fe/Ce2Sn2O7 might be a factor to further improve the reaction performance.
3.8.2 Reaction between NO+O2 and pre-adsorbed NH3 species
Like the first series of experiments, the catalysts were cleaned with the same procedure and then kept at 150 °C. As exhibited in Figure 11A1,B1, as soon as a 500 ppm NH3+Ar stream was introduced onto the samples, the adsorbed NH3 intermediates were generated, which displayed three bands at 1,599, 1,201 and 1,160 cm-1 belonging to NH3 attached on Lewis acidic sites, and other three peaks at 3,394, 3,258 and 3,150 cm-1 attributed to the vibration of N-H bonds for NH3 molecules[56,66]. Notably, on the Fe/Ce2Sn2O7 catalyst, a unique NH4+ band linked to Brønsted acidic sites appears at 1,442 cm-1, which is beneficial to the reaction[67].
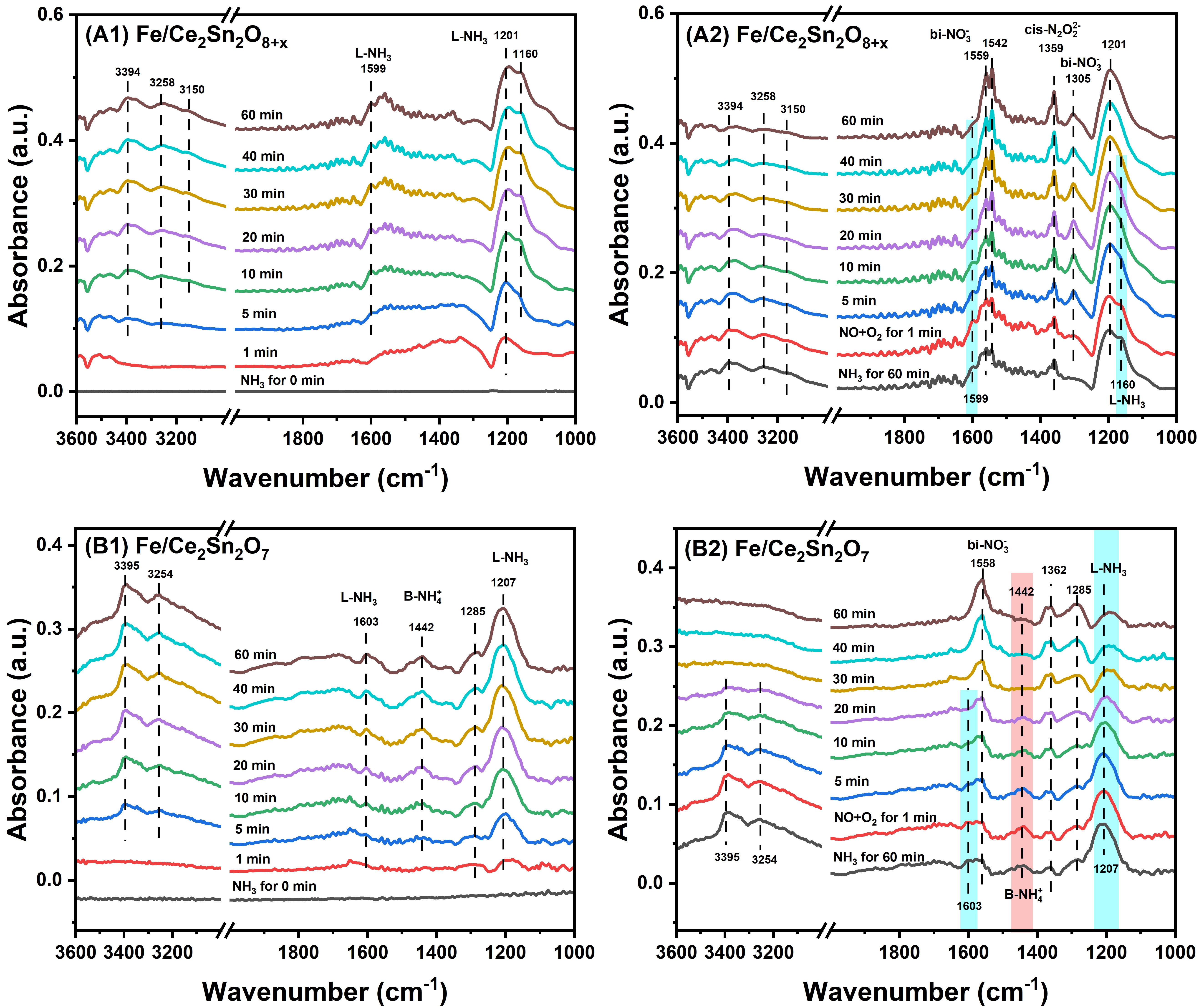
Figure 11. In-situ DRIFTS study on Fe/Ce2Sn2O8+x and Fe/Ce2Sn2O7 catalysts at 150 °C; (A1) and (B1) in a 500 ppm of NH3/Ar flow, (A2) and (B2) in a 500 ppm of NO+5% O2/Ar flow after pre-adsorbing NH3. DRIFTS: diffuse reflectance infrared Fourier transform spectroscopy.
After 40 min exposure in NH3, the catalyst surfaces were saturated, as indicated by the stabilized peak intensity. The catalysts were then flushed by a 50 mL/min Ar flow for 1 h to remove physically adsorbed NH3. Afterward, a 500 ppm NO+5 vol % O2+Ar balance flow was switched into the in-situ cell. Figure 11A2 shows that on the Fe/Ce2Sn2O8+x catalyst, three bands at 1,559, 1,542 and 1,305 cm-1 assigned to bidentate nitrate, and one peak at 1,359 cm-1 assigned to cis-N2O22- appeared rapidly in the first 1 minute[15,68]. With the increase of the exposing time, the peaks for NH3 molecules at 3,394, 3,258 and 3,150 cm-1, and NH3 attached on Lewis acidic sites at 1,599 and 1,160 cm-1 weakened until they vanished in the end[69].
Compared with the Fe/Ce2Sn2O8+x catalyst, Figure 11B2 shows that on Fe/Ce2Sn2O7, the decrease of NH4+ groups adsorbed on the Brønsted acidic sites at 1,442 cm-1 is observed as well by extending the exposing time in NO+O2. This indicates that both NH4+ and coordinated NH3 species can be involved in the reaction over Fe/Ce2Sn2O7 pyrochlore catalyst.
Summarizing these results, it is rational to conclude that NH3-SCR reaction over both Fe/Ce2Sn2O7 and Fe/Ce2Sn2O8+x catalysts follows a Langmuir-Hinshelwood mechanism, which involves the adsorption of both NOx and NH3 molecules on the surface of the catalysts during the reaction. However, on Fe/Ce2Sn2O7, additional active intermediates can be formed for both NOx and NH3 adsorption compared with Fe/Ce2Sn2O8+x, which accounts for its better NH3-SCR performance.
3.8.3 Reaction between O2 and pre-adsorbed CO species
Figure 12 displays the in-situ DRIFTS results of the transient reaction between O2 and pre-adsorbed CO on the two catalysts. As displayed in Figure 12A1, for Fe/Ce2Sn2O8+x, after 60 minutes of CO treatment, monodentate carbonate (1,548 and 1,359 cm-1), bidentate carbonate (1,650 and 1,268 cm-1), and polydentate carbonate (1,192 cm-1) were detected[70,71]. In contrast, the Fe/Ce2Sn2O7 catalyst predominantly exhibited bidentate carbonate (1,650, 1,582, and 1,236 cm-1) along with bridged carbonate (1,759 cm-1)[72,73], as shown in Figure 12B1. Meanwhile, characteristic peaks attributable to gaseous CO2 were observed for both catalysts in the range of 2,300-2,400 cm-1, indicating that lattice oxygen participated in the CO oxidation process, following the Mars-van Krevelen (MvK) mechanism.
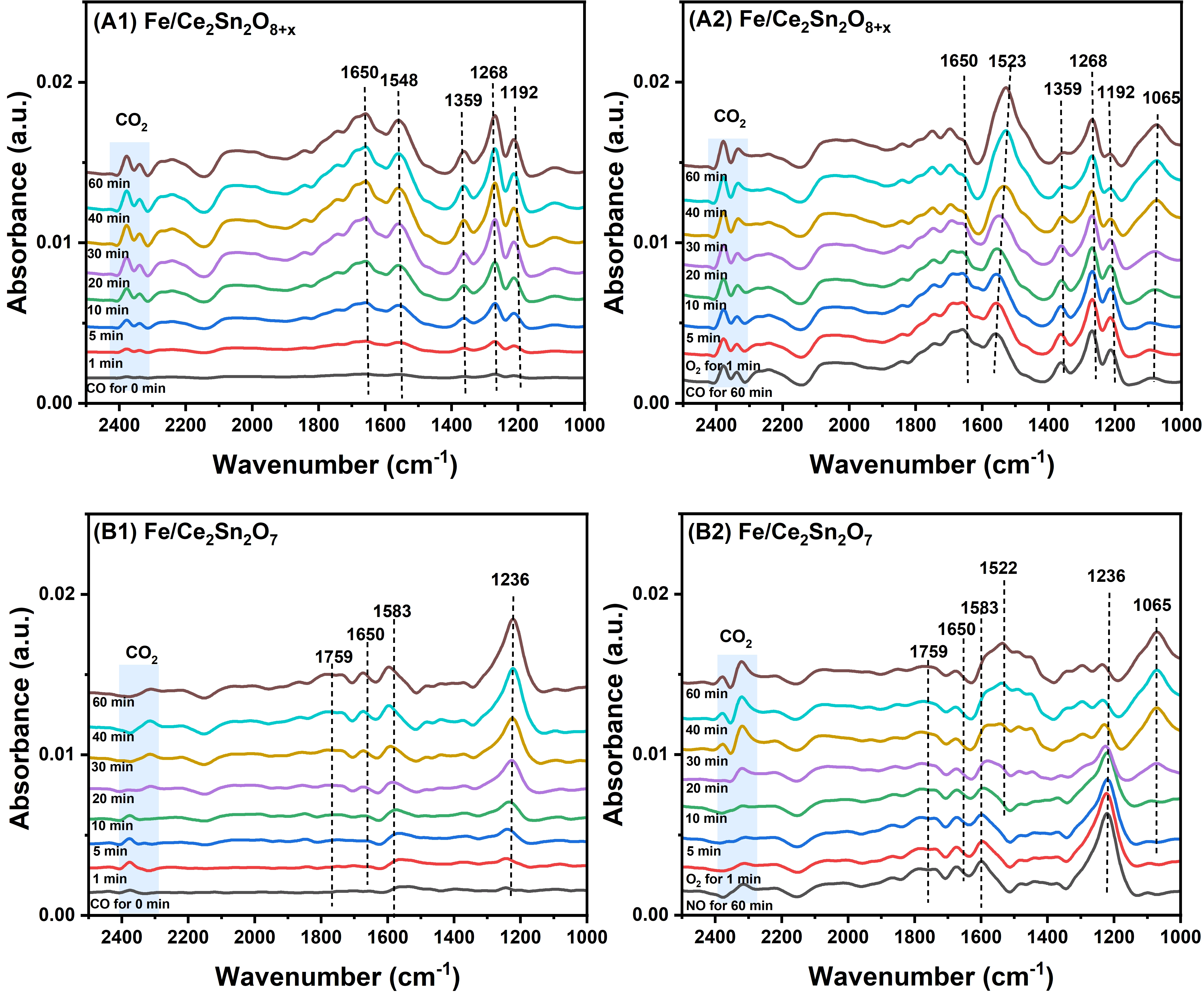
Figure 12. In-situ DRIFTS study on Fe/Ce2Sn2O8+x and Fe/Ce2Sn2O7 catalysts at 150 °C. (A1) and (B1) exposed in a 2,000 ppm CO/Ar flow, (A2) and (B2) exposed in a 5% O2/Ar flow after pre-adsorbing CO. DRIFTS: diffuse reflectance infrared Fourier transform spectroscopy.
Upon the subsequent introduction of O2, the bidentate carbonate (1,650 cm-1) and monodentate carbonate (1,359 cm-1) species adsorbed on the Fe/Ce2Sn2O8+x surface, and the bidentate carbonate (1,236 cm-1) on the Fe/Ce2Sn2O7 surface gradually diminished, accompanied by a progressive increase in the intensity of the gaseous CO2 peak (Figure 12A2,B2). This suggests that the carbonate intermediates formed with lattice oxygen sites actively participate in the CO oxidation reaction, a mark of the Mars–van Krevelen (MvK) mechanism. Remarkably, both catalysts exhibited a time-dependent intensification of the bidentate carbonate band at 1,523 cm-1[73,74]. This phenomenon can be attributed to the adsorption of O2 at oxygen vacancies upon O2 introduction, leading to the formation of peroxide species (1,065 cm-1)[75]. These O2- species subsequently react with surface carbonates, facilitating their transformation into more stable carbonates, which can ultimately promote the evolution of CO2[76]. It is noteworthy that the active bidentate carbonate species on Fe/Ce2Sn2O7 decreases more rapidly than that on Fe/Ce2Sn2O8+x, testifying that Fe/Ce2Sn2O7 is more active in CO oxidation, in line with the activity tests.
Based on the in-situ DRIFTS results (Figure 10, Figure 11 and Figure 12), we have drawn the possible reaction pathways on the Fe/Ce2Sn2O7 catalyst (Scheme 1). First, gas-phase NOx adsorbs on the catalyst surface to form active surface bidentate nitrate and bridge nitrite. Simultaneously, gas-phase NH3 adsorbs on the catalyst surface to form coordinated NH3 (B-NH4+ and L-NH3). Notably, on Fe/Ce2Sn2O7, the additional B-NH4+ ions generated on the extra Brønsted acid sites are also highly reactive, especially at low temperatures. The formed nitrate intermediates and NH3 intermediates subsequently react with each other to produce the final products, following a Langmuir-Hinshelwood mechanism.
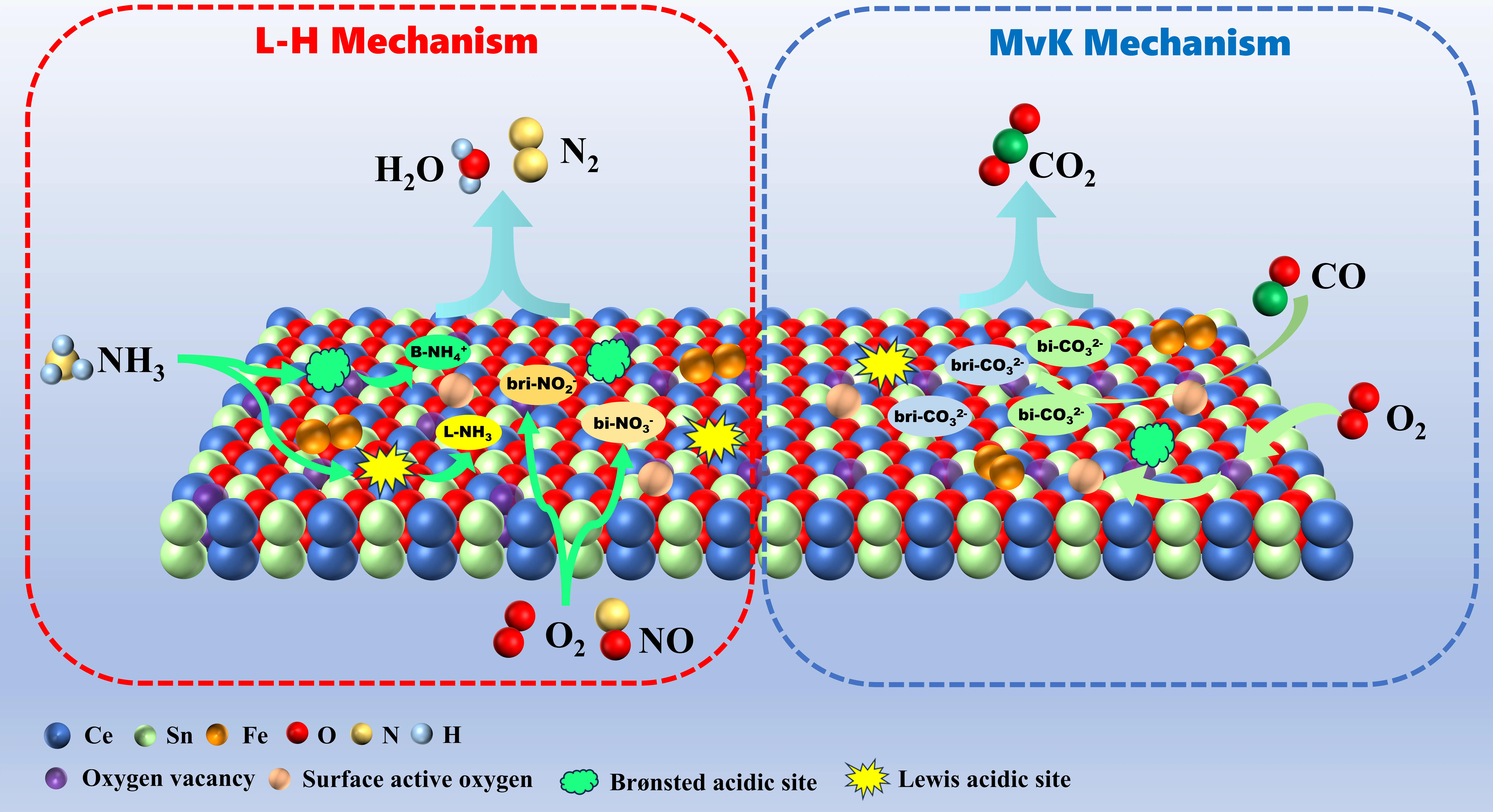
Scheme 1. Illustration of NH3-SCR and CO oxidation reaction on Fe/Ce2Sn2O7 catalysts.
The CO oxidation pathway on the surface oxygen vacancies of Fe/Ce2Sn2O7 proceeds as follows: CO reacts with adjacent surface-active oxygen to form carbonate intermediates (e.g., bridged carbonate or bidentate carbonate). These carbonate intermediates are unstable and decompose to generate CO2. In turn, gas-phase O2 molecules will adsorb onto the oxygen vacancies, and restore the consumed active oxygen species, following the MvK mechanism.
Fe/Ce2Sn2O7 uniquely generates Brønsted-bound NH4+ and bridge nitrite intermediates during NH3-SCR, which are absent on Fe/Ce2Sn2O8+x. These species accelerate the Langmuir-Hinshelwood surface reaction, leading to faster NO conversion. For CO oxidation, the higher oxygen vacancy density on Fe/Ce2Sn2O7 promotes the Mars-van Krevelen cycle, as evidenced by the faster consumption of bidentate carbonate intermediates and the stronger peroxide signal upon O2 introduction, thus improving the CO oxidation activity.
4. A Brief Discussion
In this study, to design efficient and applicable catalysts for elimination of NO and CO together, a paramorphism effect strategy has been adopted to fabricate catalysts by loading FeOx onto Ce2Sn2O7 pyrochlore and Ce2Sn2O8+x solid solutions supports. The XRD, Raman and TEM results have proven that a disordered pyrochlore phase has formed in Ce2Sn2O7 support. In contrast, mixed solid solutions with mainly Sn4+ doped into the CeO2 matrix phase and minorly Ce4+ doped into the SnO2 lattice phase are present in Ce2Sn2O8+x support. Although the metal element composition of both supports is the same, after supporting FeOx, Fe/Ce2Sn2O7 exhibits obviously better catalytic performance than Fe/Ce2Sn2O8+x. A paramorphs effect is distinctly observed, which could be a useful strategy to design catalysts not only for NH3-SCR, but also for many other catalytic reactions.
Noteworthily, in NH3-SCR reaction, the surface acidity and redox property of a catalyst are critical factors to determine its catalytic performance. As testified by EPR, more oxygen vacancies are present on Fe/Ce2Sn2O7 than on Fe/Ce2Sn2O8+x, thus it can adsorb and activate gaseous O2 molecules more easily than Fe/Ce2Sn2O8+x. DFT calculation has also confirmed that the oxygen vacancy formation energy of Fe/Ce2Sn2O7 is lower than that of Fe/Ce2Sn2O8+x. A2B2O7 pyrochlores are stoichiometric chemicals with a definite structure, which generally have superior thermal resistance, and possess structure 8a oxygen defects that can extend to the surface[34]. Naturally, Fe/Ce2Sn2O7 could possess more surface oxygen vacancies and a better capability than Fe/Ce2Sn2O8+x to activate gas phase O2. In addition, on Fe/Ce2Sn2O7, more Ce3+ cations are formed than on Fe/Ce2Sn2O8+x, which is an additional factor to promote the O2 activation. As substantiated by H2-TPR, O2-TPD and XPS O1s signals, Fe/Ce2Sn2O7 owns a richer amount of active oxygen anions (O22- and O2-) on its surface than Fe/Ce2Sn2O8+x. Figure 13A shows that the Rw and Rs on the two catalysts are positively correlated to the number of active surface oxygen sites, testifying that it is one of the crucial elements to determine the reaction performance.
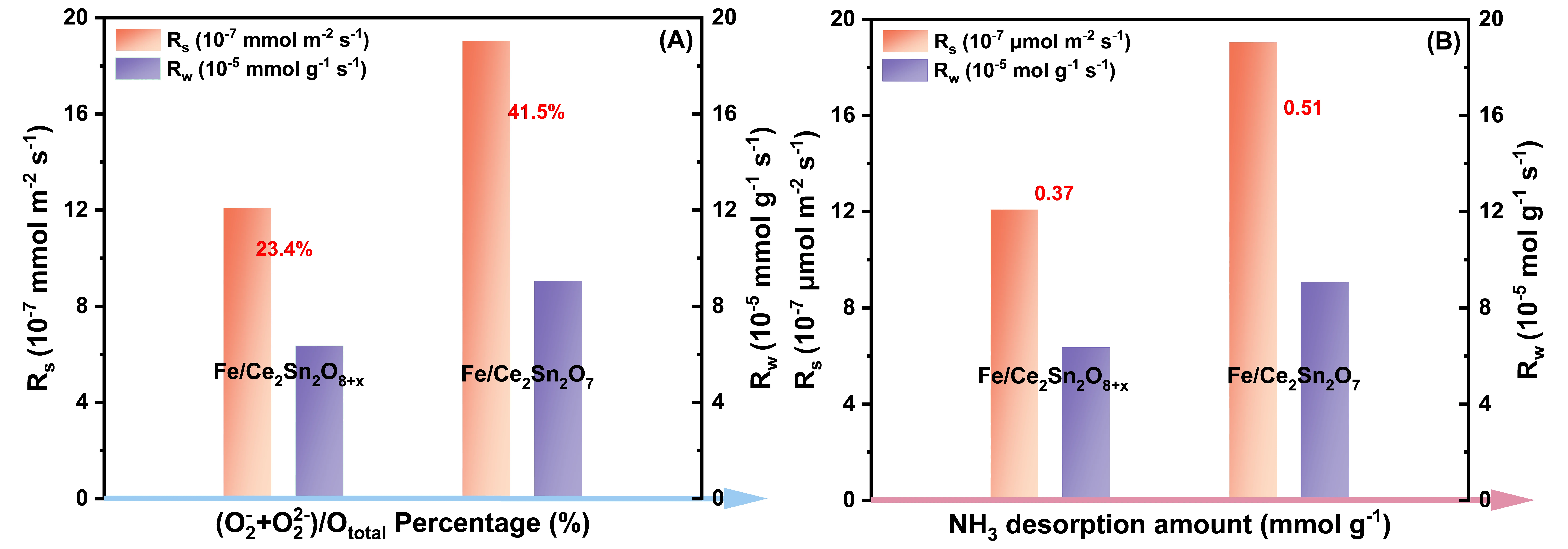
Figure 13. Rw and Rs of the catalysts at 115 °C versus (O2-+O22-)/Ototal amount (A), Rw and Rs of catalysts at 115 °C versus NH3 desorption amount.
Moreover, in NH3-SCR reaction, surface acidic sites are also of great importance, which can adsorb and activate NH3. NH3-TPD experiments have testified that a larger number of acidic sites are formed on Fe/Ce2Sn2O7 than on Fe/Ce2Sn2O8+x, which could also be closely related to the abundance of surface defects. What is more interesting, NH3 adsorption in-situ DRIFTS tests have proved that adsorbed NH4+ groups attached to Brønsted acidic sites have been uniquely formed on Fe/Ce2Sn2O7, which is in additional to the adsorbed NH3 species on Lewis acid sites found on both catalysts. It was reported that the NH4+ species can contribute to the generation of NH4NO2 or NH4NO3 intermediates to improve NH3-SCR performance at low temperatures[77]. Figure 13B shows that the Rw and Rs are also positively correlated to the number of acid sites, demonstrating that it is another crucial reason to control the reaction performance.
In terms of CO oxidation, the better performance of Fe/Ce2Sn2O7 than Fe/Ce2Sn2O8+x can be attributed to its higher concentration of surface oxygen vacancies, as evidenced by EPR and DFT calculations. These vacancies facilitate the activation of gaseous O2 into reactive oxygen species (O22- and O2-), which play a pivotal role in the Mars–van Krevelen mechanism. In-situ DRIFTS results further reveal that carbonate intermediates formed on Fe/Ce2Sn2O7 are more reactive and readily transformed into CO2 upon O2 exposure. This highlights the critical role of oxygen vacancies/reactive oxygen species in determining the activity for CO oxidation.
5. Conclusions
In this work, a paramorphism strategy has been adopted to design efficient catalysts for elimination of NO and CO together. Specifically, a Ce2Sn2O7 pyrochlore and a Ce2Sn2O8+x solid solution paramorph compounds have been successfully synthesized with a hydrothermal method to support the FeOx active component. It is discovered that Fe/Ce2Sn2O7 displays better catalytic performance than Fe/Ce2Sn2O8+x, showing an evident paramorphism effect.
As proved by XRD, Raman, TEM/HRTEM and EDS mapping tests, a disordered cubic pyrochlore phase is formed in Ce2Sn2O7 samples. In contrast, mixed solid solutions are present in Ce2Sn2O8+x samples. The loaded FeOx, which is finely dispersed in an amorphous state or small clusters, has different interactions with the two types of supports.
EPR and DFT calculations have testified that it is easier for Fe/Ce2Sn2O7 to generate more surface defects than for Fe/Ce2Sn2O8+x, thus forming a richer amount of active O22- and O2- sites. Furthermore, the number of acid sites on Fe/Ce2Sn2O7 is larger than on Fe/Ce2Sn2O8+x. As a plus, NH4+ linked to Brønsted acidic sites and bridged nitrite are uniquely formed as reactive intermediates on Fe/Ce2Sn2O7, as testified by the in-situ DRIFTS experiments. Noteworthy, Fe/Ce2Sn2O7 also exhibits better sulfur and water vapor tolerance than Fe/Ce2Sn2O8+x.
In-situ DRIFTS tests have testified that NH3-SCR reaction on both catalysts follows a Langmuir-Hinshelwood mechanism, involving the adsorption of both NO and NH3 to form reactive surface intermediates critical to the reaction. It is also demonstrated that both catalysts follow the Mars-van Krevelen mechanism for CO oxidation. The higher density of oxygen vacancies on the Fe/Ce2Sn2O7 catalyst promotes O2 activation to form O22- species, thus resulting in its better CO oxidation activity than Fe/Ce2Sn2O8+x.
Supplementary materials
The supplementary material for this article is available at: Supplementary materials.
Authors contribution
Yang Y: Data curation, formal analysis, investigation, methodology, writing-original draft, funding acquisition.
Song T: Data curation, formal analysis, investigation, methodology, visualization, writing-original draft.
Ma J, Zhang S: Investigation, formal analysis, methodology, validation.
Huang J: Investigation, formal analysis.
Shen J: Investigation, formal analysis, funding acquisition.
Xu X: Project administration, resources, software.
Xu J: Formal analysis, project administration, methodology, software, resources.
Fang X: Project administration, resources.
Wang X: Conceptualization, data curation, formal analysis, funding acquisition, validation, project administration, supervision, writing-review & editing
Conflicts of interest
The authors declare no conflicts of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
All data generated or analyzed during this study are included in this article and its supplementary information files.
Funding
The authors acknowledge deeply the financial supporting by the National Natural Science Foundation of China (Grant No. 22376090, 22362026, 22172071), the Natural Science Foundation of Jiangxi Province (Grant No. 20242BAB26029), the Key Laboratory Foundation of Jiangxi Province for Environment and Energy Catalysis (Grant No. 20242BCC32041), and the Graduate Students Innovation Special Foundation of Jiangxi Province (Grant No. YC2024-B011).
Copyright
© The Author(s) 2026.
References
-
1. Zhang R, Liu N, Lei Z, Chen B. Selective transformation of various nitrogen-containing exhaust gases toward N2 over zeolite catalysts. Chem Rev. 2016;116(6):3658-3721.[DOI]
-
2. Niu K, Liu Q, Liu C, Yu Z, Zheng Y, Su Y, et al. Unraveling the role of oxygen vacancies in metal oxides: Recent progress and perspectives in NH3-SCR for NOx removal. Chem Eng J. 2024;487:150714.[DOI]
-
3. Han L, Cai S, Gao M, Hasegawa JY, Wang P, Zhang J, et al. Selective catalytic reduction of NOx with NH3 by using novel catalysts: State of the art and future prospects. Chem Rev. 2019;119(19):10916-10976.[DOI]
-
4. Cheng K, Liu J, Zhao Z, Wei Y, Jiang G, Duan A. Direct synthesis of V–W–Ti nanoparticle catalysts for selective catalytic reduction of NO with NH3. RSC Adv. 2015;5(56):45172-45183.[DOI]
-
5. Meng D, Zhan W, Guo Y, Guo Y, Wang L, Lu G. A highly effective catalyst of Sm-MnOx for the NH3-SCR of NOx at low temperature: promotional role of Sm and its catalytic performance. ACS Catal. 2015;5(10):5973-5983.[DOI]
-
6. Wu H, Liu W, Liang Y, Liu Q. Design strategy of the MnOx catalyst for SCR of NO with NH3: Mechanism of lead poisoning and improvement method. Inorg Chem. 2023;62(42):17341-17351.[DOI]
-
7. Bian C, Luo X, Chen X, Liu R, Li J, Zhu G, et al. One-pot synthesis of Ce-SSZ-39 zeolite with performance in the NH3-SCR reaction. Inorg Chem. 2024;63(23):10798-10808.[DOI]
-
8. Liu J, Shi X, Yu Y, Zhang M, Liu D, He H. Excellent hydrocarbon tolerance of CeO2-WO3-SnO2 oxide catalyst for the NH3-SCR of NO. Appl Catal B Environ. 2023;324:122283.[DOI]
-
9. Yang Y, Zhang L, Song T, Huang Y, Xu X, Xu J, et al. On the monolayer dispersion behavior of Co3O4 on HZSM-5 support: Designing applicable catalysts for selective catalytic reduction of nitrogen oxides by ammonia. Front Chem Sci Eng. 2023;17(11):1741-1754.[DOI]
-
10. Zhang K, Luo N, Huang Z, Zhao G, Chu F, Yang R, et al. Recent advances in low-temperature NH3-SCR of NOx over Ce-based catalysts: Performance optimizations, reaction mechanisms and anti-poisoning countermeasures. Chem Eng J. 2023;476:146889.[DOI]
-
11. Huang X, Zhang K, Peng B, Wang G, Muhler M, Wang F. Ceria-based materials for thermocatalytic and photocatalytic organic synthesis. ACS Catal. 2021;11(15):9618-9678.[DOI]
-
12. Yan L, Liu Y, Zha K, Li H, Shi L, Zhang D. Deep insight into the structure–activity relationship of Nb modified SnO2–CeO2 catalysts for low-temperature selective catalytic reduction of NO by NH3. Catal Sci Technol. 2017;7(2):502-514.[DOI]
-
13. Qi G, Yang RT. Performance and kinetics study for low-temperature SCR of NO with NH3 over MnOx–CeO2 catalyst. J Catal. 2003;217(2):434-441.[DOI]
-
14. Liu Z, Feng X, Zhou Z, Feng Y, Li J. Ce-Sn binary oxide catalyst for the selective catalytic reduction of NOx by NH3. Appl Surf Sci. 2018;428:526-533.[DOI]
-
15. Liu C, Han J, Bi Y, Wang J, Guo M, Liu Q. A novel Cerium-Tin composite oxide catalyst with high SO2 tolerance for selective catalytic reduction of NOx with NH3. Catal Today. 2021;376:65-72.[DOI]
-
16. Liu J, Shi X, Lv Z, Yu Y, He H. Ceria–tungsten–tin oxide catalysts with superior regeneration capacity after sulfur poisoning for NH3-SCR process. Catal Sci Technol. 2022;12(8):2471-2481.[DOI]
-
17. Bai Y, Li H, Tong D, Niu X, Li Z, Zhu Y. Improving low-temperature NH3-SCR denitration activity and resistance to H2O and SO2 over Nb-modified NbaSn0.3CeOx catalysts by enhancing acidity to compensate for its weak oxidation ability. Sep Purif Technol. 2024;329:125212.[DOI]
-
18. Qi L, Sun Z, Yang T, Wang J, Tang Q, Huang T, et al. Effect of different introduction methods of cerium and tin on the properties of titanium-based catalysts for the selective catalytic reduction of NO by NH3. J Colloid Interface Sci. 2022;613:320-336.[DOI]
-
19. Liu J, Huo Y, Shi X, Liu Z, Shan Y, Yu Y, et al. Insight into the remarkable enhancement of NH3-SCR performance of Ce-Sn oxide catalyst by tungsten modification. Catal Today. 2023;410:36-44.[DOI]
-
20. Lai Q, Liu Y, Zhang L, Li X, Qiu Z, Xu X, etal . Expounding the monolayer dispersion threshold effect of SnO2/Beta catalysts on the selective catalytic reduction of NOx (NOx-SCR) by C3H6. Mol Catal. 2021;504:111464.[DOI]
-
21. Zhang J, Liu Y, Sun Y, Peng H, Xu X, Fang X, et al. Tetragonal Rutile SnO2 Solid Solutions for NOx-SCR by NH3: Tailoring the Surface Mobile Oxygen and Acidic Sites by Lattice Doping. Ind Eng Chem Res. 2018;57(31):10315-10326.[DOI]
-
22. Almousawi M, Xie S, Kim D, Ye K, Zhang X, Loukusa J, et al. Hydroxyls on CeO2 support promoting CuO/CeO2 catalyst for efficient CO oxidation and NO reduction by CO. Environ Sci Technol. 2024;58(1):883-894.[DOI]
-
23. Ma Y, Li Y, Liang P, Min X, Sun T. SnO2-modified CuMnOx catalysts for humid CO oxidation at low temperature. J Ind Eng Chem. 2024;138:300-310.[DOI]
-
24. Stonkus OA, Zadesenets AV, Slavinskaya EM, Stadnichenko AI, Svetlichnyi VA, Shubin YV, et al. Pd/CeO2-SnO2 catalysts with varying tin content: Promotion of catalytic properties and structure modification. Catal Commun. 2022;172:106554.[DOI]
-
25. Kardash TY, Derevyannikova EA, Slavinskaya EM, Stadnichenko AI, Maltsev VA, Zaikovskii AV, et al. Pt/CeO2 and Pt/CeSnOx catalysts for low-temperature CO oxidation prepared by plasma-arc technique. Front Chem. 2019;7:114.[DOI]
-
26. Zahir MH, Suzuki T, Fujishiro Y, Awano M. Hydrothermal synthesis of Sr–Ce–Sn–Mn–O mixed oxidic/stannate pyrochlore and its catalytic performance for NO reduction. Mater Chem Phys. 2009;116(1):273-278.[DOI]
-
27. Zhang S, Xu J, Xu Z, Li Y, Wang P, Xiao L, et al. Decoding the Reactivity Enhancement of Ln2Ce2O7 Compounds (Ln = Yb, Y, Tb, and Gd) for Soot Combustion: The Remarkable Contribution of Variable Valence A-Sites. Inorg Chem. 2024;63(15):6798-6812.[DOI]
-
28. Xu J, Xi R, Gong Y, Xu X, Liu Y, Zhong X, et al. Constructing Y2B2O7 (B = Ti, Sn, Zr, Ce) compounds to disclose the effect of surface acidity–basicity on product selectivity for oxidative coupling of methane (OCM). Inorg Chem. 2022;61(29):11419-11431.[DOI]
-
29. Zhang S, Wang P, Xu Z, Gong M, Cao J, Shen J, et al. Insights into the exceptional support phase effect on Ag/Sm2Ce2O7: Modulating Ag distribution and Ag-support interaction to construct reactive soot oxidation catalysts. J Catal. 2025;442:115893.[DOI]
-
30. Zhang J, Wang D, Lai L, Fang X, Xu J, Xu X, et al. Probing the reactivity and structure relationship of Ln2Sn2O7 (Ln = La, Pr, Sm and Y) pyrochlore catalysts for CO oxidation. Catal Today. 2019;327:168-176.[DOI]
-
31. Chen HY, Wang X, Sachtler WMH. Reduction of NOx over various Fe/zeolite catalysts. Appl Catal A Gen. 2000;194-195:159-168.[DOI]
-
32. Zhang W, Shi X, Yan Z, Shan Y, Zhu Y, Yu Y, et al. Design of high-performance iron–niobium composite oxide catalysts for NH3-SCR: Insights into the interaction between Fe and Nb. ACS Catal. 2021;11(15):9825-9836.[DOI]
-
33. Han L, Gao M, Feng C, Shi L, Zhang D. Fe2O3–CeO2@Al2O3 nanoarrays on Al-mesh as SO2-tolerant monolith catalysts for NOx reduction by NH3. Environ Sci Technol. 2019;53(10):5946-5956.[DOI]
-
34. Xu J, Xi R, Xu X, Zhang Y, Feng X, Fang X, et al. A2B2O7 pyrochlore compounds: A category of potential materials for clean energy and environment protection catalysis. J Rare Earths. 2020;38(8):840-849.[DOI]
-
35. Zhang S, Xu J, Lu C, Ouyang R, Ma J, Zhong X, et al. Preparation method investigation and structure identification by XRD and Raman techniques for A2B2O7 composite oxides. J Am Ceram Soc. 2024;107(5):3475-3496.[DOI]
-
36. Batzill M, Diebold U. The surface and materials science of tin oxide. Prog Surf Sci. 2005;79(2-4):47-154.[DOI]
-
37. Bie Y, Li T, Li F. Hydrothermal synthesis of Ce2Sn2O7 nanoparticles for effective sonocatalytic performance. Ceram Int. 2023;49(14):22726-22735.[DOI]
-
38. Khan MS, Ameer H, Chi Y. Label-free and ultrasensitive electrochemiluminescent immunosensor based on novel luminophores of Ce2Sn2O7 nanocubes. Anal Chem. 2021;93(7):3618-3625.[DOI]
-
39. Ganesan M, Jayaraman V, Selvaraj P, Mani KM, Kim DH. Pyrochlore cerium stannate (Ce2Sn2O7) for highly sensitive NO2 gas sensing at room temperature. Appl Surf Sci. 2023;624:157135.[DOI]
-
40. Xu X, Tong Y, Zhang J, Fang X, Xu J, Liu F, et al. Investigation of lattice capacity effect on Cu2+-doped SnO2 solid solution catalysts to promote reaction performance toward NOx-SCR with NH3. Chin J Catal. 2020;41(5):877-888.[DOI]
-
41. Jayaraman V, Palanivel B, Ayappan C, Chellamuthu M, Mani A. CdZnS solid solution supported Ce2Sn2O7 pyrochlore photocatalyst that proves to be an efficient candidate towards the removal of organic pollutants. Sep Purif Technol. 2019;224:405-420.[DOI]
-
42. Gupta HC, Brown S, Rani N, Gohel VB. A lattice dynamical investigation of the Raman and the infrared frequencies of the cubic A2Sn2O7 pyrochlores. Int J Inorg Mater. 2001;3(7):983-986.[DOI]
-
43. Vandenborre MT, Husson E, Chatry JP, Michel D. Rare-earth titanates and stannates of pyrochlore structure; vibrational spectra and force fields. J Raman Spectrosc. 1983;14(2):63-71.[DOI]
-
44. Xu X, Liu F, Tian J, Peng H, Liu W, Fang X, et al. Modifying the surface of γ-Al2O3 with Y2Sn2O7 pyrochlore: Monolayer dispersion behaviour of composite oxides. ChemPhysChem. 2017;18(12):1533-1540.[DOI]
-
45. Wu T, Zhang P, Wei Y, Xiong J, Han D, Li T, et al. Surface oxygen vacancies induced by calcium substitution in macroporous La2Ce2–xCaxO7–δ catalysts for boosting low-temperature oxidative coupling of methane. ACS Catal. 2024;14(3):1882-1902.[DOI]
-
46. Wang Y, Chen L, Wang W, Wang X, Li B, Zhang S, et al. Revealing the excellent low-temperature activity of the Fe1–xCexOδ-S catalyst for NH3-SCR: Improvement of the lattice oxygen mobility. ACS Appl Mater Interfaces. 2023;15(14):17834-17847.[DOI]
-
47. Xu X, Liu X, Ma L, Liang N, Yang S, Liu H, et al. Construction of surface synergetic oxygen vacancies on CuMn2O4 spinel for enhancing NO reduction with CO. ACS Catal. 2024;14(5):3028-3040.[DOI]
-
48. Ye L, Lu P, Yan X, Huang H. Boosting simultaneous catalytic removal of NOx and toluene via cooperation of Lewis acid and oxygen vacancies. Appl Catal B Environ. 2023;331:122696.[DOI]
-
49. Yang J, Hu S, Fang Y, Hoang S, Li L, Yang W, et al. Oxygen vacancy promoted O2 activation over perovskite oxide for low-temperature CO oxidation. ACS Catal. 2019;9(11):9751-9763.[DOI]
-
50. Zhang J, Fan Y, Chen L, Yang L, Zhou L, Luo X, et al. Promoting the catalytic activity and SO2 resistance of CeO2 by Ti-doping for low-temperature NH3-SCR: Increasing surface activity and constructing Ce3+ sites. Chem Eng J. 2023;473:145272.[DOI]
-
51. Zhao W, Shen M, Zhu Y, Ren X, Li X. Insights into synergy of copper and acid sites for selective catalytic reduction of NO with ammonia over zeolite catalysts. Catalysts. 2023;13(2):301.[DOI]
-
52. Zhang N, Wang J, Li Q, Xin Y, Zheng L, Wang Y, et al. Enhanced selective catalytic reduction of NO with NH3 over homoatomic dinuclear sites in defective α-Fe2O3. Chem Eng J. 2021;426:131845.[DOI]
-
53. Zhang N, Tong J, Miyazaki S, Zhao S, Kubota H, Jing Y, et al. Mechanism of NH3-SCR over P/CeO2 catalysts investigated by operando spectroscopies. Environ Sci Technol. 2023;57(43):16289-16295.[DOI]
-
54. Yasumura S, Qian Y, Kato T, Mine S, Toyao T, Maeno Z, et al. In situ/operando spectroscopic studies on the NH3–SCR mechanism over Fe–zeolites. ACS Catal. 2022;12(16):9983-9993.[DOI]
-
55. Dambournet D, Demourgues A, Martineau C, Durand E, Majimel J, Vimont A, et al. Structural investigations and acidic properties of high surface area pyrochlore aluminium hydroxyfluoride. J Mater Chem. 2008;18(21):2483.[DOI]
-
56. Mo D, Qin Q, Huang C, Tao L, Li C, Qiu J, et al. Regulating the distribution of iron active sites on γ-Fe2O3 via Mn-modified α-Fe2O3 for NH3-SCR. Appl Catal B Environ Energy. 2024;349:123869.[DOI]
-
57. Xue H, Guo X, Meng T, Guo Q, Mao D, Wang S. Cu-ZSM-5 catalyst impregnated with Mn–co oxide for the selected catalytic reduction of NO: Physicochemical property–catalytic activity relationship and in situ DRIFTS study for the reaction mechanism. ACS Catal. 2021;11(13):7702-7718.[DOI]
-
58. Wu Y, Li G, Chu B, Dong L, Tong Z, He H, et al. NO reduction by CO over highly active and stable perovskite oxide catalysts La0.8Ce0.2M0.25Co0.75O3 (M = Cu, Mn, Fe): Effect of the role in B site. Ind Eng Chem Res. 2018;57(46):15670-15682.[DOI]
-
59. Chung CH, Tu FY, Chiu TA, Wu TT, Yu WY. Critical roles of surface oxygen vacancy in heterogeneous catalysis over ceria-based materials: A selected review. Chem Lett. 2021;50(5):856-865.[DOI]
-
60. Yi Y, Liu H, Chu B, Qin Z, Dong L, He H, et al. Catalytic removal NO by CO over LaNi0.5M0.5O3 (M = Co, Mn, Cu) perovskite oxide catalysts: Tune surface chemical composition to improve N2 selectivity. Chem Eng J. 2019;369:511-521.[DOI]
-
61. Liu X, Huang L, Wu X, Wang Z, Dong G, Wang C, et al. Bi2Zr2O7 nanoparticles synthesized by soft-templated sol-gel methods for visible-light-driven catalytic degradation of tetracycline. Chemosphere. 2018;210:424-432.[DOI]
-
62. Gou Z, Huang C, Zhou G, Ren X, Deng L, Wang T, et al. Coupling and electronic synergistic effects of Fe/CeO2 composite to achieve high efficiency and selectivity for RWGS reaction. J CO2 Util. 2024;81:102728.[DOI]
-
63. Wang Y, Zhang G, Xi Y, Tang Z, Feng H. Elucidating the electron confinement effect on CeFeW/ZrO2 catalysts to enhance SO2 resistance in the low temperature NH3-SCR reaction. Sep Purif Technol. 2024;346:127569.[DOI]
-
64. Hu H, Cai S, Li H, Huang L, Shi L, Zhang D. Mechanistic aspects of deNOx processing over TiO2 supported co–Mn oxide catalysts: Structure–activity relationships and in situ DRIFTs analysis. ACS Catal. 2015;5(10):6069-6077.[DOI]
-
65. Jiang M, Yan Z, Zhang Y, Zhang C, Chang C, Xiao M, et al. Simultaneous modification of redox and acidic properties of FeOx catalysts derived from MIL-100(Fe) via HPW incorporation for NH3-SCR. Appl Catal B Environ Energy. 2024;358:124416.[DOI]
-
66. Fang Z, Zhang S, Wang A, Guo Y, Guo Y, Wang L, et al. Er-modified MnOx for selective catalytic reduction of NOx with NH3 at low temperature: Promoting effect of erbium on catalytic performance. J Rare Earths. 2023;41(6):917-925.[DOI]
-
67. Chen L, Wang Y, Wang X, Wang Q, Li B, Li S, et al. Brønsted acid enhanced hexagonal cerium phosphate for the selective catalytic reduction of NO with NH3: in situ DRIFTS and DFT investigation. J Hazard Mater. 2022;424:127334.[DOI]
-
68. Zhang T, Shi T, Wang Y, Hao Y, Gao Y, Li H, et al. Orchestrating dual adsorption sites and unravelling Ce-Mn interaction and reaction mechanisms for efficient NH3-SCR. J Catal. 2024;429:115260.[DOI]
-
69. Chen Y, Li J, Teng W, Wu H, Liu W, Ren S, et al. Revealing the effect of electronic interaction between dual redox sites on CeO2-Fe2O3 NH3-SCR catalyst through interface contact tailoring. Fuel. 2023;353:129179.[DOI]
-
70. Yang S, Ren S, Yang H, Li X, Zheng C, Li X, et al. Morphology regulation of CuO/CeO2 catalyst for simultaneous NH3-SCR denitration and CO oxidation. J Environ Chem Eng. 2025;13(5):118868.[DOI]
-
71. Shen Z, Xing X, Guo P, Ren S, Wei D, Meng H. Insight into the Cd resistance mechanism of MoCuMnOx/CeO2 catalyst for simultaneously NH3-SCR denitration and CO oxidation: Directional capture of Cd by MoOx. Chem Eng J. 2026;527:171626.[DOI]
-
72. He Y, Liu J, Zhang G, Zang P, Li G, Wang Y. Insights into the structure-activity relationships of highly efficient CuCe oxides for the low temperature CO oxidation and CO-SCR. J Energy Inst. 2022;104:142-155.[DOI]
-
73. Lv D, Liu J, Zhang G, Wang Y, Zhao Y, Li G. Novel insights for simultaneous NOx and CO Removal: Cu+-Sm3+-Ov-Ti4+ asymmetric active site promoting NH3-SCR coupled with CO oxidation reaction. Chem Eng J. 2024;481:148534.[DOI]
-
74. Jia B, Liu J, Kang J, Zhang G, Lv D, Wang Y. Investigating NH3-SCR coupled with CO oxidation reaction mechanisms on titanium nanotube-loaded CuMnFe composite metal catalysts. Appl Surf Sci. 2024;652:159299.[DOI]
-
75. Zhang S, Wang P, Yang Y, Tang Y, Wu Z, Fang X, et al. Rich-defect Ru–Tb2Ce2O7+x catalysts for highly efficient soot particulate elimination: Insights into the key roles of Ru–O–Ce/Tb interfacial bonds, Ru0 sites, and surface vacancies. Environ Sci Technol. 2026;60(8): 6427-6441. [ DOI:10.1021/acs.est.5c14990][DOI]
-
76. Lustemberg PG, Yang C, Wang Y, Ganduglia-Pirovano MV, Wöll C. Synergistic effects in low-temperature CO oxidation on cerium oxide surfaces. J Am Chem Soc. 2025;147(8):6958-6965.[DOI]
-
77. Wang Y, Zhu B, Sin S, Zhang Z, Tan C, Gu Z, et al. Lattice oxygen activation triggered by ultrasonic shock significantly improves NO selective catalytic reduction. ACS Catal. 2024;14(12):9265-9274.[DOI]
Copyright
© The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Publisher’s Note
Share And Cite