Abstract
With global warming, urban workers engaged in physically demanding occupations are increasingly exposed to severe heat stress. While the physical health risks are well recognized, less is known about how heat stress affects cognitive function at the neurophysiological level, an understanding critical for protecting worker well-being. This study conducted a controlled laboratory experiment to examine cognitive and neural responses to heat stress in middle-aged heat-exposed manual workers. Twenty participants (mid-40s) completed six representative cognitive tasks under four wet-bulb globe temperature (WBGT) conditions (23, 26, 28.5, and 31 °C). Electroencephalogram (EEG) recordings were used to monitor brain activity and assess changes in relative band power across δ, θ, α, and β frequency bands. The results revealed that performance in the executive- memory dimension peaked under moderate heat (forming an inverted-U pattern), while performance in the sensorimotor reactivity dimension remained stable or improved with rising WBGT. Neurophysiologically, rising heat stress led to increased δ band power but decreased α, θ, and β band powers. These EEG band power changes showed a nonlinear relationship with cognitive performance across both dimensions, with the left frontal cortex demonstrating higher sensitivity. Furthermore, topographical coupling maps indicated that executive-memory demanding tasks activated a more widespread cortical region than sensorimotor-reactivity demanding tasks. These findings show that graded heat stress alters brain dynamics and cognitive performance in a cognitive load-dependent manner, offering insights for designing adaptive work environments and real-time cognitive performance monitoring devices for heat- exposed workers.
Keywords
1. Introduction
The increasing prominence of global climate change is driving more frequent extreme heat events, a critical climate issue for urban areas worldwide. During summer, the urban heat island effect[1-3], dramatically intensified by dense building structures and human activities, leads to significantly higher temperatures in urban centers[4,5]. Under such extreme conditions, manual workers in heat-exposed industries such as construction and manufacturing are consistently subjected to severe heat stress[6]. This heat stress, that is a consequence of the combined burden of excessive environmental heat and the metabolic heat generated by strenuous labor, not only causes discomfort but also significantly amplifies these workers’ health and safety risks[7-9].
Nowadays, the importance of examining the impacts of the thermal environment on cognitive performance is increasingly emphasized for several compelling reasons. Firstly, the World Health Organization’s (WHO) definition of health encompasses physical, mental, and social well-being, underscoring the integral role of cognitive function in overall health[10]. Secondly, modern urban industry places significant cognitive demands on workers, requiring sustained attention, memory, sound judgment, and effective decision-making[11,12]. For instance, heat-induced cognitive fatigue among construction workers operating heavy machinery may increase the likelihood of lapses in attention or judgment, resulting in costly mistakes or potentially fatal accidents. The heat exposure regulations that rely solely on physiological thresholds inadequately address the substantial safety and efficiency risks stemming from heat-induced cognitive impairment[13-15]. Therefore, a dedicated investigation of how heat stress affects the cognitive functions of manual workers, through the critical lens of “cognitive safety”, is necessary. Understanding and mitigating these cognitive decrements associated with heat stress is essential for optimizing workplace design and establishing evidence-based heat exposure limits that truly safeguard worker well-being and operational efficiency.
Existing theories, such as “arousal theory” and the “maximal adaptability model”, provide a solid grounding for understanding how heat stress impacts cognition performance[13,16]. Building on foundational frameworks, a wealth of empirical studies has delineated the precise thermal thresholds, measured in terms of core and skin body temperature, wet-bulb globe temperature (WBGT), or effective temperature (ET), beyond which specific cognitive functions begin to deteriorate[16-18]. However, these studies fall short of revealing the immediate neural underpinnings of heat stress.
In recent years, advances in neurocognitive science and EEG hardware have made electroencephalography a widely accessible tool for probing cortex electrophysiological activity. Electroencephalogram (EEG) device captures scalp potential and neural oscillations, providing a direct window into subjects’ neurophysiological responses and cognitive function[19-21]. It promises to “open the black box” of brain activity, revealing the immediate neural mechanisms through which heat stress impairs cognition[22]. Some pioneering studies have used EEG to examine how thermal environments are associated with changes in brain activity during cognitive tasks. For instance, Lang et al.[23] exposed participants to extreme heat during a low-intensity task and observed a marked decline in relative β-band power at 39 °C, signaling impaired cognitive performance. Likewise, Choi et al.[24] evaluated attention under warmer conditions (PMV +2 and +3) and found that higher temperatures corresponded with significantly lower attention levels, as reflected in EEG markers. Niu et al.[25] examined how intense localized radiant heat affects cognition and EEG. They found that increasing localized radiant heat initially enhanced cognitive performance, α- and β-band power, vigilance, and frontal EEG asymmetry, but beyond a certain threshold, all measures declined, forming an inverted-U response.
These studies offer preliminary, yet valuable, insights into the effects of heat stress on both cognitive function and neurophysiological activity. However, these findings are far from conclusive and necessitate further extensive research to validate them and elucidate the underlying mechanisms in greater depth. Crucially, the populations examined in prior research have often been limited to young, indoor office workers, revealing a significant gap in our knowledge, particularly with regard to middle-aged individuals engaged in heat-exposed manual occupations, such as construction, manufacturing, and sanitation work. Middle-aged workers constitute the majority of the labor force in these heat-exposed sectors[26]. In comparison with younger employees, they usually have longer occupational histories and higher levels of heat acclimatization, which may influence both their physiological and cognitive responses to thermal stress[27]. Focusing on this demographic is thus demographically representative and, in addition, provides a practical basis for understanding heat stress impacts under real-world conditions.
Therefore, this study aims to bridge this critical gap by investigating the effects of varying heat stress levels on the cognitive performance of middle-aged (mid-40s) workers with heat-exposed manual occupations. The research will specifically examine their subjective thermal comfort, objective cognitive task performance, and underlying brain activity, with particular emphasis on identifying thermally sensitive cortical regions as measured by EEG, seeking to validate and provide a deeper understanding of heat stress effects on this vital, yet understudied, demographic. Specifically, this research will address the following questions:
RQ1. How does graded heat stress influence workers’ subjective thermal comfort and various cognitive task performance?
RQ2. What is the tendency in workers’ cognitive performance metrics, relative EEG band powers, and their interrelationship as heat stress intensifies?
RQ3. Which cortex regions demonstrate a higher sensitivity to heat-stress-induced cognitive performance fluctuations?
2. Literature Review
2.1 Theories and empirical evidence
2.1.1 Theoretical frameworks for explaining the relationship between human heat stress and cognition
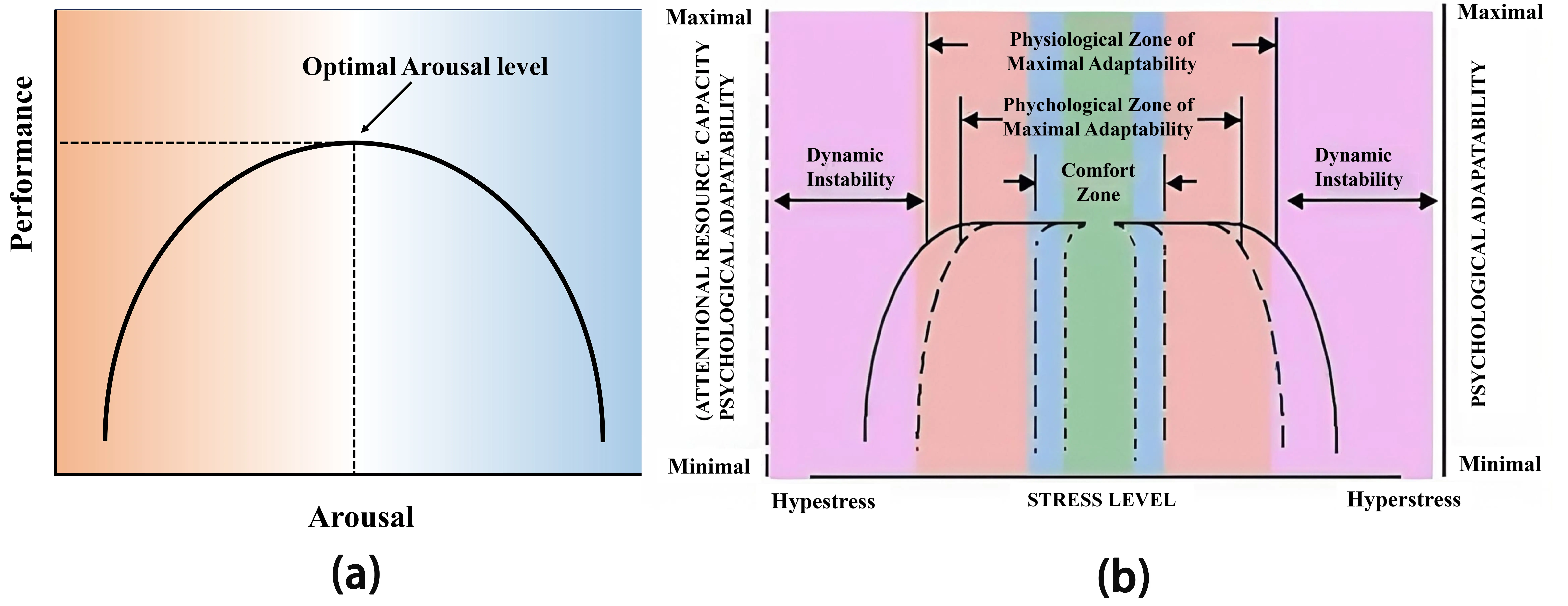
Arousal theory, embodied in the Yerkes–Dodson law, remains the most widely applied theory to describe how heat stress affects cognitive performance[28], proposing an inverted–U relationship between the level of arousal and task performance as shown in Figure 1a. Building upon arousal theory, Hancock et al.[16,29] introduced the maximal adaptability model, which offers a more detailed perspective on the effects of heat stress on cognitive performance. As depicted in Figure 1b, input stress ranges from low to high. Optimal cognitive function occurs in the normative zone, surrounded by a comfort zone where minor adjustments suffice. Increased heat leads to a psychological zone, involving adaptive tactics against mild resource depletion. In the physiological zone, performance plateaus and then declines as compensation fails. Severe heat leads to instability, homeostatic breakdown, and rapid cognitive deterioration. These theoretical models provide a coherent framework for understanding how heat stress impacts human performance and serve as valuable guides for relevant experimental design.
2.1.2 Empirical investigation of the effects of heat stress on cognitive performance
A substantial body of empirical research has affirmed the general principles put forth by these theories and models regarding the impact of heat stress on cognitive performance. However, these studies have also highlighted that temperature thresholds for optimal versus impaired performance are not universal and vary by task type and exposure conditions[30-32].
In an early study, Grether[30] categorized cognitive activities into five types: time estimation, reaction time, tracking, vigilance, and aiming. The results indicated that optimal performance occurred at approximately 26.7 °C WBGT for vigilance tasks and 29.5 °C WBGT for the others. Similarly, Ramsey and Morrissey[31] developed “isodecrement” curves that map specific temperature-time combinations onto the probability of performance decline for mental (e.g., coding, mental arithmetic), tracking, reaction time, vigilance, and memory tasks, emphasizing that task differentiation is essential and that decrements develop gradually rather than abruptly. Extending these findings, Ramsey and Kwon[32] synthesized over 150 studies and confirmed that simple mental and perceptual-motor tasks show little or even positive effects during brief exposures, while more demanding perceptual-motor tasks consistently exhibit significant performance losses at 30-33 °C WBGT.
This extensive body of work collectively underscores the complex interplay between environmental heat, task demands, and human cognitive resilience, establishing critical insights while also revealing the inherent variability that future research needs to further unravel through the precise stratification of thermal stress gradients and the strategic selection of cognitive tasks.
2.2 EEG insights into the effect of heat stress on cognition performance
2.2.1 Methodological approaches and key EEG metric
While existing theoretical and empirical studies describe when and how heat stress degrades cognitive performance, they often fall short of explaining the underlying neural mechanisms. EEG helps to address this gap by providing an objective window into how heat stress affects cognition. To capture these neurophysiological responses, researchers utilize diverse EEG devices, ranging from high-fidelity medical-grade devices (e.g., B-Alert X10/X24[33], Biosemi[34]) to more portable platforms ideal for in-situ monitoring (e.g., Emotiv EPOC[35]).
Typical analysis pipelines for EEG data begin with stringent preprocessing steps, including band-pass filtering, epoching, baseline correction, and artifact rejection[25,36-39]. The clean signal is typically decomposed into canonical frequency bands (δ, θ, α, β), and their cognitive performance correlates are summarized in Table 1. Analysis approaches, such as event-related potentials (ERP)[42], time-frequency analysis, frequency domain analysis[23,25,35-37], and connectivity analysis[38], are employed. Among these methods, power spectral density (PSD) remains a predominant measure in thermal-cognition research; it yields absolute power and relative power (band power normalized by total power)[25,37]. Furthermore, the analysis extends to the ratio of band power across either the entire set of electrodes or specific, localized electrode sites, providing a more granular understanding of neural activity[35,43].
| Band | Frequency range | Cognitive Performance Correlates[39-41] |
| δ (Delta) | 0.5-4 Hz | Reflects deep sleep or cognitive suppression; often elevated during drowsiness |
| θ (Theta) | 4-8 Hz | Associated with working memory load, alertness, and cognitive control |
| α (Alpha) | 8-13 Hz | Linked to relaxed wakefulness and inhibitory control during attentional tasks |
| β (Beta) | 13-30 Hz | Related to active thinking, focus, and problem solving |
EEG: electroencephalogram.
2.2.2 Fluctuations in EEG spectral band power under thermal stress
Among the limited but growing body of thermal-cognition studies, relative power is frequently adopted. This is primarily because it allows for straightforward comparisons of neural activity across different groups of participants[44,45]. Table 2 presents a synthesis of findings from several key studies, including sample size, thermal environmental conditions, associated EEG band power variations, the involvement of sensitive brain regions, and the impact on cognitive performance.
| Reference | Device used | Sample | Band-power tendency | Sensitive Area | Cognitive Performance |
| Lang et al.[23] | 14-ch cap | N = 48 (24 male/24 female) Age ≈ 20 | WBGT: 26, 39 °C δ: ↑ θ: ↓ α: ↑ β: ↓ | Frontal, Temporal, Occipital | ↓ Highest atWBGT = 26 °C |
| Silvia et al.[49] | MUSE 2headband | N = 23 (9 male/14 female) Age: 27 -32 | Tα: 16, 24, 31 °C θ: ↓ β: ∩ | Frontal lobe (AF8) | − |
| Niu et al.[25] | Open BCI | N = 10 (10 male/0 female) Age: 21.4 ± 1.9 | TSV: 1 to 3 θ: → α: ↑ β: ∩ | The left frontal lobe (AF3, F7, F3) | Reaction time: ↑ Accuracy: ∩ |
| Erkan. et al.[46] | Neurosky MindWave | N = 180 (90 male/90 female) Age: 19-40 | Tα: 15, 22, 30 °C δ: ↑ Θ: → α: ∩ β: ∩ | Frontal lobe | − |
| Sun et al.[50] | Emotiv EPOC X | N = 14 (7 male/7 female) Age: 21-24 | Tα: 18, 24, 20 °C θ: ∪ | Frontal lobe | − |
| Li et al.[37] | EMOTIV EPOC X | N = 30 (15 male/15 female) Age: 20-27 | Tα: 22, 26, 30 °C β: ∩ | Frontal lobe (AF3/AF4, F3/F4) | ∩ Highest at 26 °C |
| Li et al.[47] | Emotiv EPOC X | N = 28 (14 male/14 female) Age: 20-30 | TSV: -3 to +3 α: ∩ β: ∩ | Frontal (AF3, AF4, F3, F4, FC6, +(+T7, O2) | ∩ Highest at TSV = -1 to 0 |
| Kim et al.[35] | EMOTIV EPOC + | N = 20 (16 male/4 female) Age: 23-32 | TSV: -3 to 3 β: ∩ | Frontal lobe | Learning performance:∩ |
EEG: electroencephalogram; WBGT: wet-bulb globe temperature; TSV: thermal sensation vote; N: sample size, Band-power tendency symbols; ↑: increase; ↓: decrease; →: no clear change/stable; ∩: inverted-U pattern; ∪: U-shaped pattern.
Across studies, a rough tendency is observed. δ band power tends to rise with uncomfortable temperatures and is associated with poorer cognitive performance. For instance, Erkan et al.[46] observed that relative δ power was elevated, and cognitive performance was impaired, at both 15 °C and 30 °C when compared to 22 °C. Similarly, Lang et al.[23] reported that exposure to extreme heat (39 °C) led to a significant increase in whole-brain δ-band relative power, accompanied by a marked decline in accuracy on cognitive tests. In contrast, θ band power often shows a descending trend as thermal conditions shift from comfortable to uncomfortable. For instance, Niu et al.[25] observed a slight decline in relative θ band power as occupants’ thermal sensation vote (TSV) increased from 1 (slightly warm) to 3 (very hot). For α and β band power, if the environment becomes uncomfortable, α and β band power tend to decline. This pattern has been documented in studies by Erkan et al.[46], and Li et al.[37], Li et al.[47].
However, it is notable that some studies also suggest inconsistent tendencies of these band powers. For example, Niu et al.[25] showed a stable δ-band relative power as thermal comfort changed thermal comfort vote (TCV). Furthermore, the specific degree of change also varies significantly. Shin et al.[48] found only a small increase in β power (0.02-0.03) at 22.5 °C compared to 25 °C and 35 °C. Conversely, Lan et al.[43] observed much larger β power boosts (0.28-6.41 µV2) and better performance at 17 °C compared with 21 °C and 28 °C. These inconsistent findings are likely attributable to methodological heterogeneity across studies. In particular, many experiments select temperature setpoints not anchored to a standardized heat-stress index (e.g., WBGT), and there is limited harmonization in exposure definition, exposure duration, and task requirements.
2.2.3 Heat stress sensitivity across cortical regions
Beyond overall power shifts, researchers utilize head topographical maps to identify cortical regions where heat stress-related brain power changes more sensitively impact cognitive ability, offering refined spatial insights.
Li. et al.[37] demonstrated that δ showed more observable changes in the frontal and central brain regions. Similarly, Erkan et al.[46] investigated the spatial distribution of δ, θ, α, and β band power changes across different indoor temperatures. They reported that the increase in δ power activity related to fatigue was often concentrated in the frontal regions. Lang et al.[23] examined the effects of heat stress on cognitive function and brain activity by analyzing EEG band power at 14 electrodes. Their results typically indicated that the increase in δ and decrease in θ power associated with decreased cognitive performance under heat stress were more significant in frontal and midline frontal regions, while α and β power declines were predominantly observed in temporal and occipital areas. Li et al.[47] reported a broader cortical involvement in thermally induced cognitive impairment, noting band changes not only in frontal–central locations (AF3, AF4, F3, F4, FC6) but also in temporal (T7) and occipital (O2) regions.
To sum up, these studies highlight the frontal lobes’ particular vulnerability to temperature-driven EEG alterations while also implicating temporal and occipital cortices in heat-induced cognitive impairment. This distributed pattern of band-power change underscores the necessity for in-depth research into region-specific neural mechanisms underlying thermal–cognitive interactions.
2.3 Research gaps and study positioning
While prior research has identified consistent trends, such as shifts in δ, θ, α, and β relative band power and their sensitive cortical regions under thermal load, these findings still require further validation[23,25,35-37]. First, the majority of investigations have targeted younger cohorts (aged 20-30), such as students and white-collar workers, leaving workers with heat-exposed manual occupations, a group routinely exposed to extreme heat, underexplored. Second, researchers often evaluate cognitive performance using a composite score, which obscures the identification of distinct EEG band-power fluctuation patterns across different dimensions of cognitive performance. Third, the predominant use of linear correlation analyses to associate EEG band- power fluctuations with cognitive performance is inherently inadequate, as it fails to capture the complex nonlinear interactions between neural activity and cognitive performance under heat stress. The key research gaps and the positioning of the present study are summarized in Table 3.
| Dimension | Prior studies | Limitations | Present study |
| Study population | Predominantly younger cohorts (aged 20-30 years) | Underexplored in workers with heat-exposed manual occupations | Heat-exposed middle-aged manual workers (mid-40s) |
| Cognitive performance evaluation | Evaluating using a composite score | Obscures EEG band power patterns across performance dimensions | Diverse cognitive tasks and specific dimensions of performance |
| EEG–cognition association | Linear correlation analyses | Inadequate for capturing nonlinear neural–performance interactions | Applies nonlinear coupling analysis |
EEG: electroencephalogram.
This study empirically examines how EEG relative band power and sensitive cortical regions reflect the impact of heat stress on cognitive performance among heat-exposed manual workers. By employing diverse cognitive tasks and advanced nonlinear coupling analyses, it aims to reveal how neural oscillations are coupled with specific dimensions of performance under heat stress. The findings will provide neuroscientific evidence to inform targeted occupational health strategies for preventing heat-induced cognitive impairment in thermally demanding environments.
3. Methodologies
3.1 Experimental design and execution
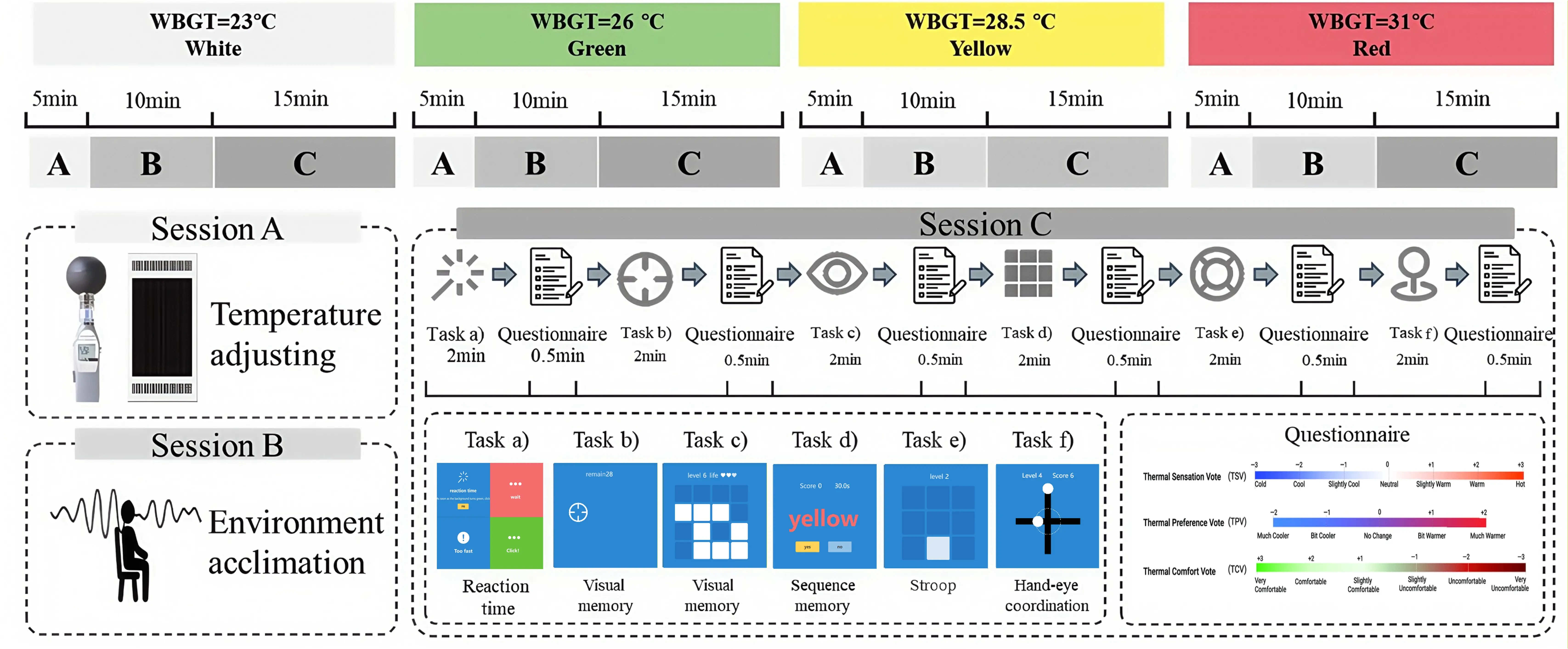
The overview of the experimental procedure of this study is shown in Figure 2. The entire experiment lasts for 120 minutes and consists of four rounds, each lasting 30 minutes and corresponding to the four target WBGT conditions. Each round consisted of three continuous sessions: Session A involved pre-adjusting the environmental chamber to the designated WBGT conditions to ensure a stable thermal environment; Session B served as an acclimatization period during which participants entered the chamber to adapt to the heat and were fitted with the EEG headset. This standardized 10-min acclimation minimized short-term thermal carryover between WBGT conditions and allowed brief physiological stabilization before EEG recording and task testing, consistent with common climate-chamber protocols[51]; Session C was dedicated to the execution of cognitive tasks and subjective questionnaires while simultaneously acquiring real-time EEG data to capture neural oscillations under the specific thermal stress. For participant safety, four environmental conditions were selected in accordance with the WBGT heat-stress risk categories specified by ISO 7243[52]. WBGT was computed according to Equation 1[53]. Low risk level (white, WBGT = 23 °C), medium risk level (green, WBGT = 26 °C), high risk level (yellow, WBGT = 28.5 °C) and very high risk level (red, WBGT = 31 °C), ensuring that no excessive heat stress was experienced. Table 4 provides the full set of measured environmental parameters, including relative humidity, wet-bulb temperature, globe temperature, and air temperature, for each risk level. Environmental parameters at each WBGT at condition were configured to align with representative local climate characteristics. The entire experimental protocol was approved by the Ethics Committee of the Hong Kong Polytechnic University.
| Wet-Bulb Globe Temperature (WBGT °C) | Relative Humidity (RH %) | Wet-BulbTemperature (Twb °C) | Globe Temperature (Tg °C) | Air temperature (Tα °C) |
| 23.0 ± 0.3 | 60 ± 1 | 21.7 ± 0.4 | 26.1 ± 0.3 | 25.8 ± 0.5 |
| 26.0 ± 0.4 | 61 ± 1 | 24.6 ± 0.4 | 29.5 ± 0.4 | 29.0 ± 0.4 |
| 28.5 ± 0.4 | 61 ± 1 | 27.0 ± 0.3 | 32.5 ± 0.5 | 31.2 ± 0.3 |
| 31.0 ± 0.3 | 62 ± 1 | 28.9 ± 0.3 | 36.3 ± 0.5 | 35.3 ± 0.4 |
WBGT: wet-bulb globe temperature.
Where, Twb is the wet-bulb temperature; Tg is the globe temperature; Ta is the air temperature.
3.1.1 Experimental instruments and setting
A purpose-built, thermally insulated cabin was used to provide a controlled environment for precise manipulation of heat stress levels and was situated within a temperature-controlled laboratory to ensure experimental stability and safety. The size of this cabin was 0.9 m (length)× 0.9 m (width)× 1.9 m (height). A 0.5 m × 0.4 m viewing window was opened on one side of the cabin, permitting participants to see the computer screen and use a mouse to perform cognitive tasks and complete subjective questionnaires. To simulate the thermal radiation environment of workers, an adjustable electric radiation heating panel with a maximum power of 1,800 W was installed on the top of the cabin, serving as a heat source that mimics the radiant heat exposure commonly encountered by heat-exposed manual workers. At the same time, an 8788AZ Portable WBGT meter was used in the experiment to monitor WBGT temperature in real time. This device was located in the central part of the cabin, close to the subjects, with a height of 1.2 m, corresponding to the heart area of the seated participants. The detailed experimental set up is shown in Figure 3.
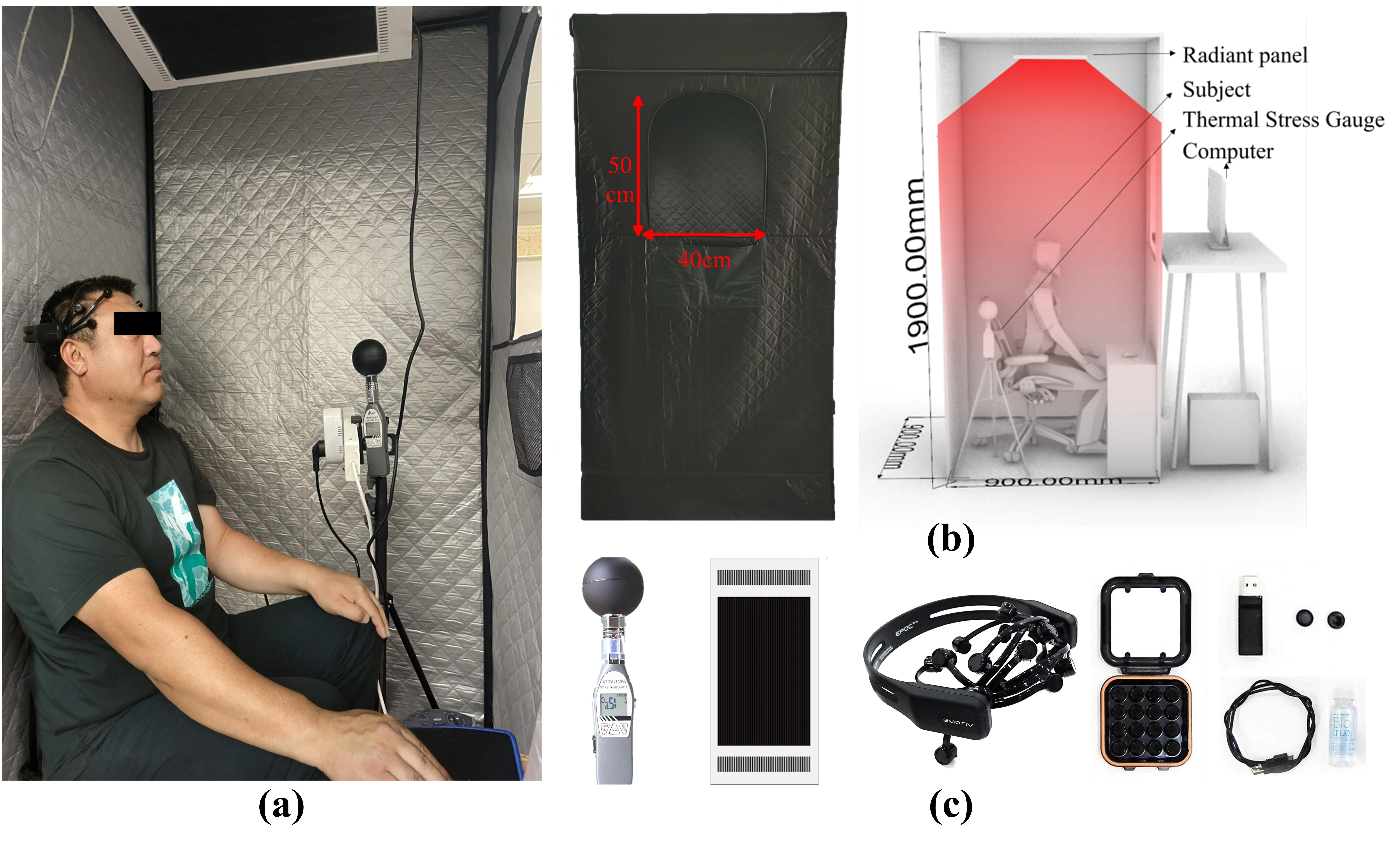
Figure 3. Experimental set up (a) The participant in the cabin; (b) The setup in the cabin; (c) Experiment equipment.
During the experiment, the Emotiv EPOC+ device was utilized to collect real-time EEG signals from the subjects, enabling the continuous acquisition of their scalp oscillation data. This device is a high-resolution, non-invasive and portable wireless headset. The device comprises 14 channels: AF3, F7, F3, FC5, T7, P7, O1, O2, P8, T8, FC6, F4, F8, and AF4, in accordance with the international 10–20 electrode placement system. Common model sense (CMS) and driven right leg (DRL) are the reference electrodes behind the ear for signal baseline calibration. The detailed instrument information and other environmental parameters involved in the experiment is listed in Table 5.
| Equipment | 8788 AZ Portable WBGT Monitor | Emotiv EPOC+ | Radiation Heating PanelFSD15-4000 |
| Range | WBGT: 0 to 50 °C Air Temperature: -10 to 70 °C Humidity: 0 to 100% RH | 14-channel EEG Wireless; headset; EEG signal amplitude: ±400 μV Frequency range: 0.2-43 Hz | Heating power: 1,800 W Operating temperature: 0-300 °C Operating Voltage: 220-380 V Resistance at room temperature: 20 Ω Overall dimensions: 700 × 390 × 60 mm |
| Accuracy | Temperature: ±0.5 °C Humidity: ±3% RH (at 25 °C, 10-90% RH) WBGT: ±0.5 °C | Resolution: 14-bit (1.95 μV per bit) Sampling rate: 128 Hz (256 Hz optional) |
WBGT: wet-bulb globe temperature; RH: relative humidity.
3.1.2 Participants
In this study, with reference to prior relevant literature, a total of 20 workers were enrolled (10 males and 10 females). Inclusion criteria were as follows: (1) employment as heat-exposed middle-aged manual workers; (2) residency in the study locale for a minimum of five years; (3) demonstrated proficiency with the experimental procedures following standardized training[54]; (4) absence of neurological disorders or recent use of medications known to affect cognitive function; and (5) normal vision and hearing. The participants had a mean age of 45.2 ± 1.6 years. Female participants had a mean weight of 65.7 ± 3.1 kg and a mean height of 164.7 ± 1.1 cm, whereas male participants averaged 78.7 ± 3.0 kg in weight and 175.3 ± 1.7 cm in height. The combined body mass index (BMI) for all participants was 24.9 ± 0.3 kg/m2, as listed in Table 6.
| Characteristic | Total (N = 20) | Male (N = 10) | Female (N = 10) |
| Age (years) | 45.2 ± 1.6 | 46.2 ± 0.9 | 44.2 ± 1.1 |
| Body Weight (kg) | 72.2 ± 3.6 | 78.7 ± 3.0 | 65.7 ± 3.1 |
| Height (cm) | 170.0 ± 3.1 | 175.3 ± 1.7 | 164.7 ± 1.1 |
| BMI (kg/m2) | 24.9 ± 0.3 | 25.6 ± 0.3 | 24.2 ± 0.3 |
BMI: body mass index.
All participants received a detailed briefing on the study protocol and provided written informed consent prior to participation. Before each round, participants were allowed to rest freely (e.g., use the washroom, access drinking water). Experimental sessions were consistently scheduled within a fixed time window (3:00-5:00 p.m.). To ensure participant safety and well-being, subjects were permitted to withdraw from the experiment at any point if they felt uncomfortable due to the environmental conditions.
3.1.3 Cognitive performance test
Six cognitive tasks were selected from Human Benchmark[55], a web platform for brain games and cognitive tests – to evaluate participants’ cognitive performance. Each task was designed to closely align with an established paradigm in cognitive and human-performance research, thereby providing a standard behavioral index for the targeted cognitive function. To suit the target population, all tasks relied exclusively on mouse interactions rather than keyboard input, as many workers have limited typing experience. The tasks were deliberately selected for their straightforward and intuitive design, which facilitated quick familiarization by the participants. A structured training session with repeated practice was conducted prior to data collection to support task familiarity and minimize practice effects. To minimize fatigue and cumulative exhaustion, each set of cognitive tasks was limited to 15 minutes. Participants remained in the climate- controlled cabin, observing the test interface through a window. Tasks could be repeated within the allotted time, and the mean score across trials was used for analysis. To enable direct comparison of performance across different tasks, all scores were Z-score standardized (Equation 2), thereby removing discrepancies in units or scales. The specific content of these six tasks is introduced below.
Where,
Task a) -Reaction time test: A colored rectangle is displayed on the screen. When the color changes from red to green, participants need to click the screen as soon as possible. This process is repeated five times. The system records the time interval from each color change to the click and calculates the average reaction time as the final result. This task measures Simple Reaction Time, which is widely used to assess basic response speed[56].
Task b) -Visual memory: A 3 × 3 grid is displayed on the screen, and then several squares are randomly illuminated. The number of illuminated squares and the size of the grid increase with each level. Participants need to remember the illuminated positions. During the answering stage, participants click on the correct position. After making three mistakes, the highest level reached will be recorded. This task is a variation of the Visual Patterns Test, designed to assess the capacity of the visual short-term memory component of the working memory system[57].
Task c) -Aiming training: 30 target points are randomly generated on the screen. Participants use the mouse to quickly click on each target. The system records the average inter-click interval. This task aligns with the Fitts’ Law paradigm, evaluating visuomotor processing speed and responsiveness to dynamic spatial targets[58].
Task d) -Stroop Task: Color words are displayed on the screen. Participants judge whether the font color is consistent with the text content and choose “Yes” or “No”. 1 point is added for each correct answer. The highest score is recorded within 30 seconds or after an error occurs. This task utilizes the classic Stroop Interference paradigm to measure selective attention and cognitive control ability[59].
Task e) -Sequence memory: The squares at different positions on the screen light up successively. Participants click in sequence. The initial sequence length is one. After each pass, the level is upgraded and the length increases by one. After an error occurs, the highest level is recorded. This task adapts the classic Corsi Block-Tapping paradigm to measure visuospatial working memory[60].
Task f) -Hand-eye coordination task: The screen displays a target area and two moving small balls. Participants need to click the mouse when the small balls enter the target area to make them stay. Starting with low speed and a large target area, the difficulty progressively increases until the test terminates after two consecutive errors. Employing a Target Interception paradigm, this task assesses sustained attention and dynamic visuomotor coordination[61].
3.1.4 Questionnaire and survey
The subjective questionnaire comprised two parts: the first part collected basic demographic information (e.g., participants’ names, height, weight, age); the second part focuses on the subjective evaluation of the thermal environment, utilizing three common thermal comfort metrics: TSV, TCV, and thermal preference vote (TPV). As shown in Figure 4, TSV employed a Likert 7-point scale (-3 = very cold to +3 = very hot, with 0 = neutral). TPV used a five-point scale (-2 = prefer much colder to +2 = prefer much hotter, with 0 = no preference). TCV utilized a seven-point scale (-3 = very uncomfortable to +3 = very comfortable, with 0 = no feeling). After completing each cognitive task, the thermal comfort survey was presented via the computer interface, and data were automatically recorded in the back-end database.
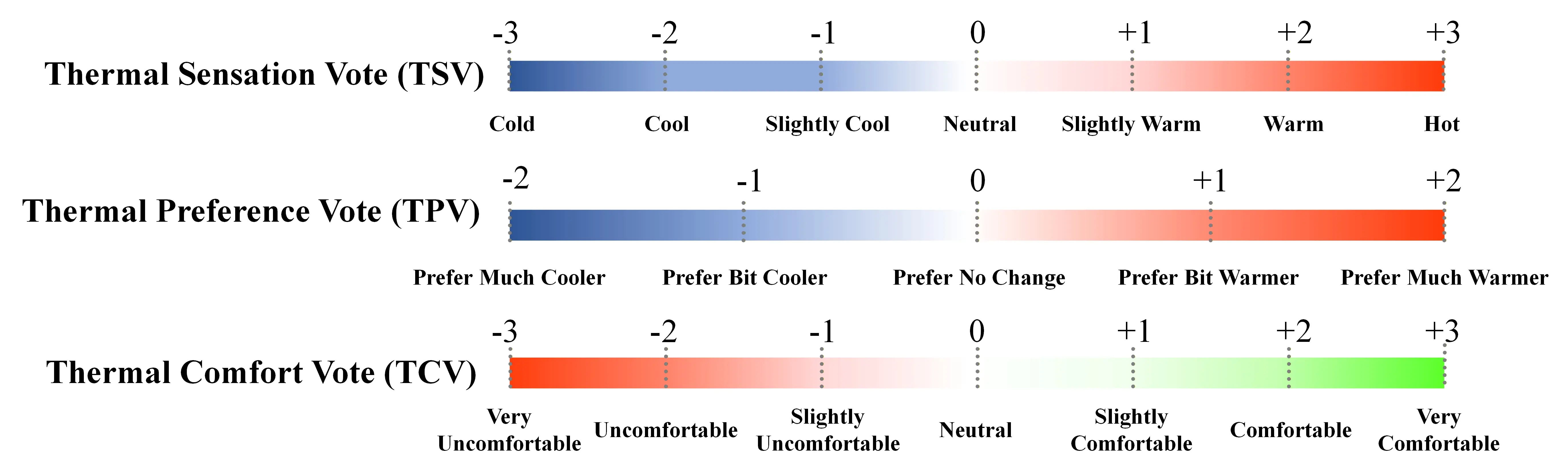
Figure 4. The scale of thermal comfort metrics (TSV, TPV, and TCV). TSV: thermal sensation vote; TPV: thermal preference vote; TCV: thermal comfort vote.
3.2 EEG data preprocessing
Due to their weak nature and susceptibility to noise interference, EEG signals require preprocessing to extract meaningful data from raw recordings[39,62,63]. This crucial step, guided by standard EEG manual handbooks, involved four procedures implemented using the EEGLAB plugin in MATLAB software (Figure 5)[63]. These steps collectively aim to maximally eliminate non-brain- derived artifacts while preserving genuine neural oscillation components. Finally, the extracted 1-30 Hz band frequency EEG data were retained for subsequent analysis.
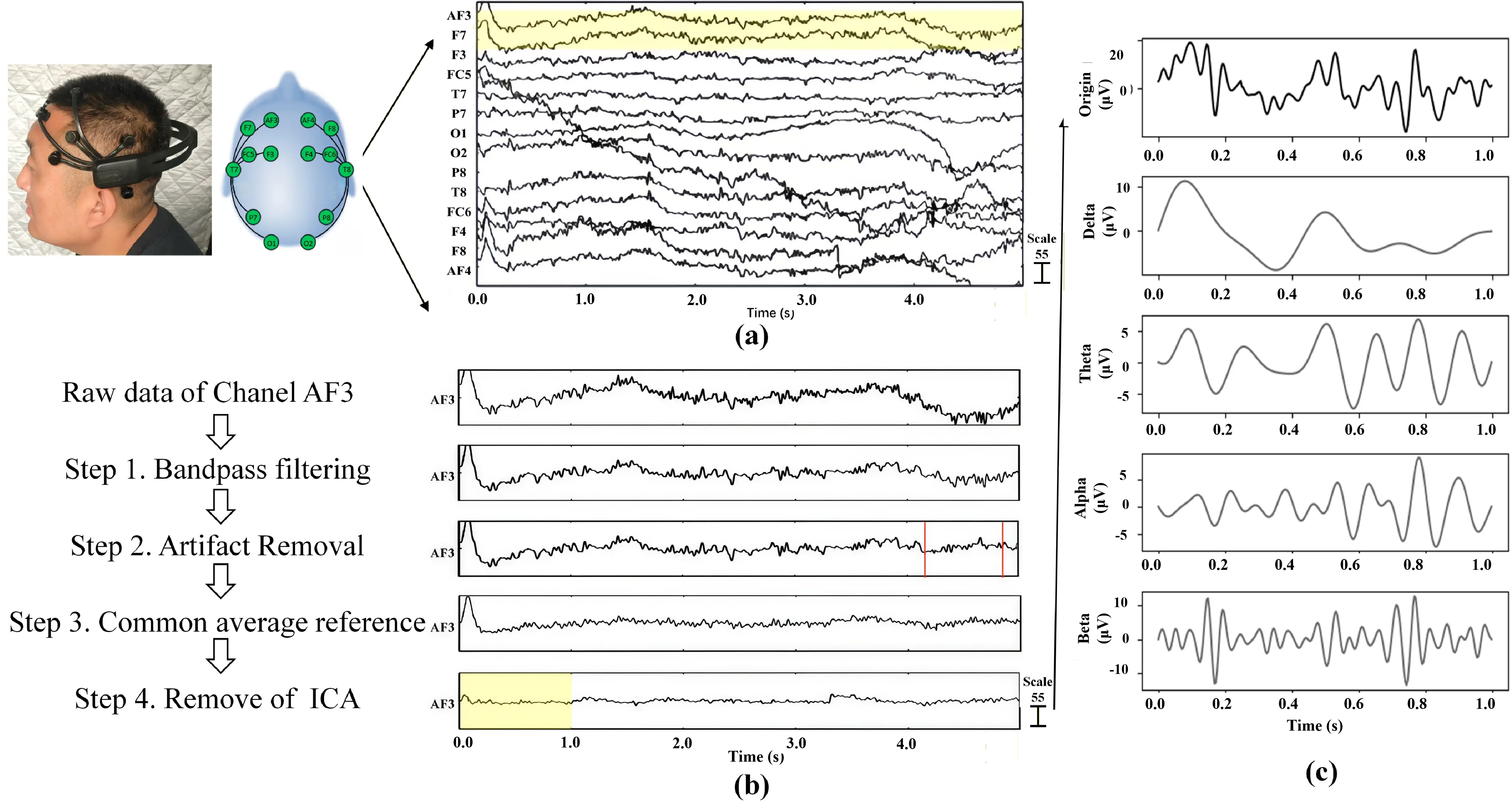
Figure 5. EEG data pre-processing (a) Raw EEG data; (b) Step in EEG data process; (c) Band frequency. EEG: electroencephalogram.
3.2.1 Bandpass filtering
The raw EEG data underwent bandpass filtering (1-30 Hz). This process, using fast Fourier transform (FFT), converts the signal from the time domain to the frequency domain. It specifically enables the retention of brainwave frequencies relevant to cognitive function while effectively filtering out low-frequency drift (e.g., from breathing or heartbeat artifacts) and high-frequency noise (e.g., electromyographic interference).
3.2.2 Artifact subspace reconstruction (ASR)
ASR is a component-based artifact removal method for removing transient or large-amplitude artifacts in multi-channel EEG recordings. The preprocessing workflow began with a rigorous channel cleaning procedure: electrodes were identified as defective and removed if they remained flat for more than 5 seconds, exhibited a high-frequency noise standard deviation exceeding 4, or showed a correlation coefficient lower than 0.8 with neighboring channels. Subsequently, ASR was applied to the remaining data by constructing a statistical model of the signal. Using a sliding 500ms window, the algorithm identified abnormal components exceeding a standard deviation threshold of 10. This process enabled the reconstruction of clean EEG signals while preserving the integrity of neural activity, ensuring high-quality data for subsequent neurophysiological analysis.
3.2.3 Common average reference (CAR)
CAR is a re-referencing technique where the average potential of all electrodes serves as the reference. By subtracting this global average from each channel’s potential, CAR aims to mitigate common-mode noise and improve the signal’s spatial resolution, predicated on the assumption of a diffuse electric field distribution.
3.2.4 Independent component analysis (ICA) for artifact removal
ICA decomposes EEG data into statistically independent components, facilitating the isolation and removal of non-neuronal signals like electrooculogram (EOG) and electromyogram (EMG) artifacts. To ensure a rigorous artifact rejection process, a manual inspection of each component was conducted. Components were identified and rejected as artifacts (e.g., blinks or myogenic activity) if they exhibited a classification probability for Muscle or Eye ranging from 0.9 to 1.0 via the EEGLAB plugin in MATLAB, while simultaneously displaying distinct time and frequency features, as well as spatial topographies. The remaining components were then used to reconstruct a clean and high-quality EEG signal for subsequent neurophysiological analysis.
3.3 EEG relative band power calculation
The relative band power was the primary metric for characterizing brain cortical oscillatory activity. The relative-power minimizes inter-subject variability and facilitates cross-channel and group-level comparisons[25,64]. To achieve this, PSD was initially estimated for each channel’s preprocessed EEG data via the FFT using Welch’s method. Specificity, it involved segmenting the signal into overlapping windows, computing each periodogram, and then averaging them for a more stable, lower-variance spectral density estimate (Equation 3)[39,63]. Next, the absolute power Pi of a given frequency band was computed by summing the PSD across all frequencies in that band (Equation 4). To quantify the relative contribution of each band, δ, θ, α, and β, each band’s power was then normalized by the total power across these four bands (Equation 5). Figure 6 summarizes the pipeline for computing Pi in AF3 channel, alongside a topography example depicting four bands relative power across all 14 channels.
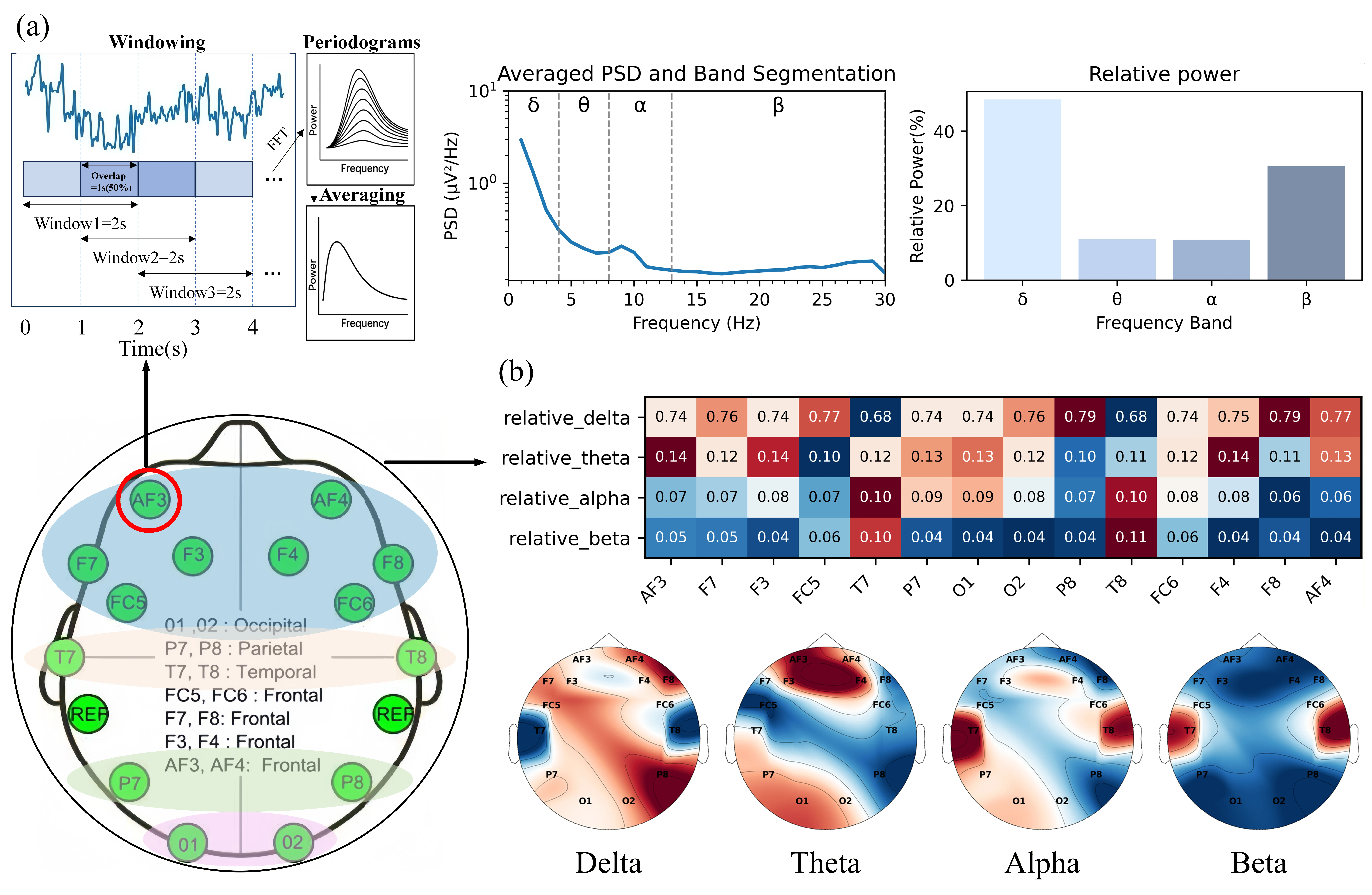
Figure 6. Relative power calculation and mapping (a) Relative power calculation of a channel; (b) 14 channels calculation and mapping. PSD: power spectral density.
Where, S(tk) is the amplitude of the kth sample, ΔT = 1/fs is the sampling interval, and N is the total number of samples.
4. Result
4.1 Group comparison analyses under four heat stress level
4.1.1 Subjective thermal comfort distributions - thermal comfort survey results
Figure 7 illustrates the overall distribution of three subjective thermal metrics, TSV, TPV, and TCV, across four WBGT levels. At 23 °C, most people feel thermally neutral (TSV = 0), and there was a strong preference for no change in thermal conditions (TPV = 0). The TCV reached its peak at +2, suggesting that the majority of people felt comfortable. As the WBGT rises to 26 °C, the TSV shifts towards 0 to 1 (neutral to slightly warm). Concurrently, the TPV indicates a preference for slightly cooler conditions (-1 to 0), while the TCV indicates slight comfort to comfort (+1 to +2). Under high-stress conditions (28.5 °C), the highest density of TSV moves to +1 (slightly warm). The TPV distinctly peaked at -1, signifying a desire for cooler conditions, and the highest density of TCV dropped to -1 (slightly uncomfortable). Finally, at 31 °C, the TSV exceeded +2 (hot). The TPV was concentrated between –1 and -2, indicating a strong preference for cooling, and the TCV reaches its maximum at -2 (uncomfortable).
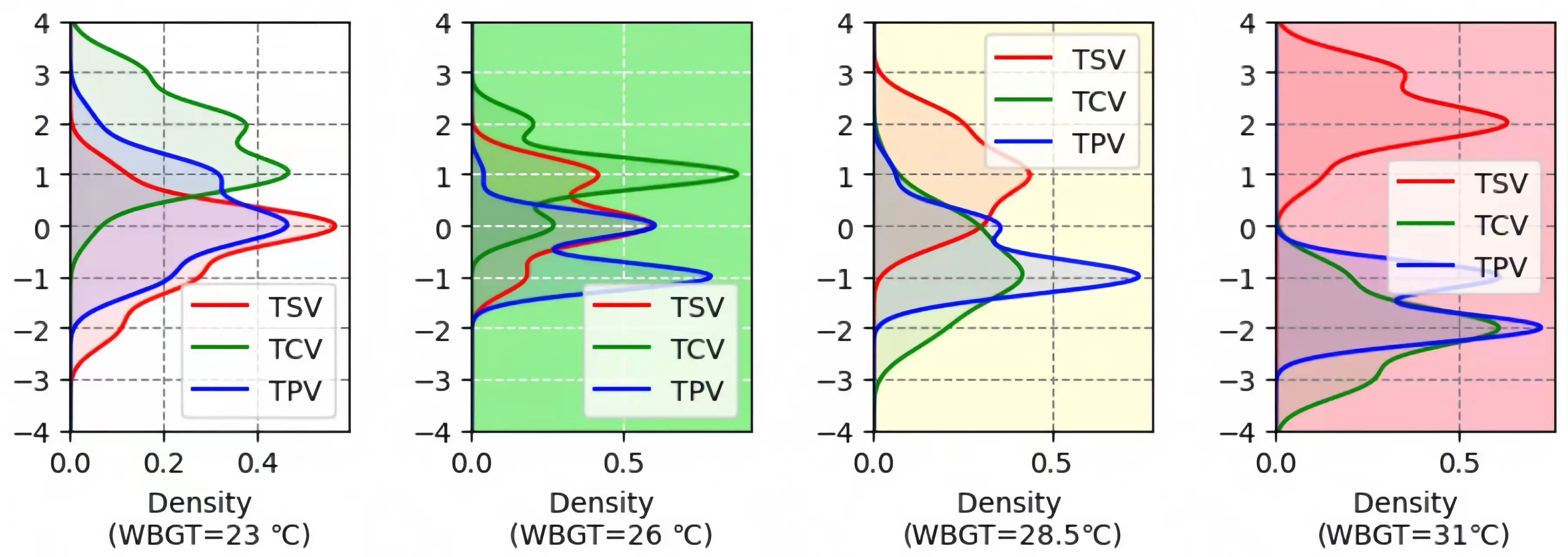
Figure 7. Subjective thermal comfort distributions across four WBGT levels. WBGT: wet-bulb globe temperature.
4.1.2 Cognitive performance patterns - cognitive tasks results
The cognitive performance of participants on six tasks under four WBGT levels is depicted in Figure 8a. The results reveal two distinct response patterns. Workers’ cognitive performance in “aiming training”, “reaction time”, and “hand-eye coordination” tasks increased monotonically with rising WBGT levels – particularly for the first one. In contrast, their cognitive performance in “visual memory”, “Stroop”, and “sequence memory” tasks followed an inverted-U pattern: cognitive efficiency peaks at 26 °C or 28.5 °C, followed by a decline at 31 °C.
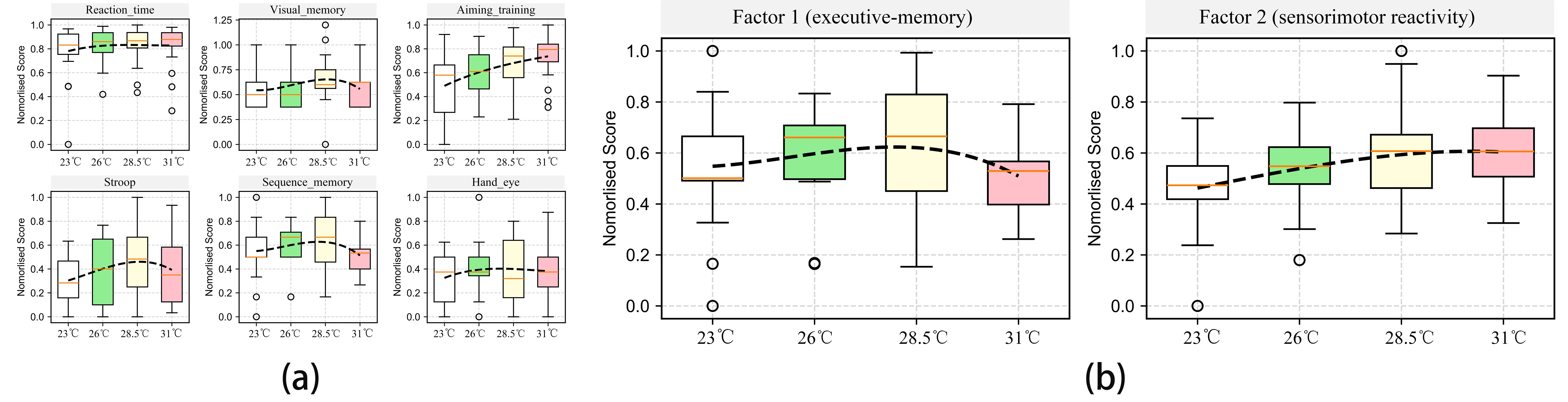
Figure 8. Distribution cognitive performance under four temperature conditions (a) Performance on six cognitive tests under four temperature conditions; (b) Performance across two dimensions under four temperature conditions.
To explore the latent structure underlying these cognitive tasks, an exploratory factor analysis (EFA) was conducted. As shown in Table 7, the Kaiser–Meyer–Olkin (KMO) index reached 0.734, indicating that the dataset was suitable for EFA. Furthermore, Bartlett’s test of sphericity was highly significant (χ2 = 754.5, p < 0.001), confirming sufficient intercorrelation among the variables[21]. The factor analysis revealed two components with eigenvalues greater than 1. Factor 1, accounting for 35.6% of the total variance, showed high loadings on “sequence memory” (0.899), “Stroop” (0.805), and “visual memory” (0.689), which indicate a latent dimension associated with cognitive control and memory. Factor 2, which explained an additional 22.5% of the variance (cumulative variance = 58.1%), was strongly associated with “reaction time” (0.638), “aiming training” (0.607), and “hand-eye coordination” (0.653). It reflects a dimension primarily related to reactivity. For interpretive clarity, Factor 1 was labeled the “executive-memory” dimension, and Factor 2 the “sensorimotor reactivity” dimension.
| Indictors | Factor1 | Factor2 | Statistical |
| Reaction time | 0.233 | 0.638 | KMO = 0.734, Bartlett χ2 = 754.5, p < 0.001 |
| Visual memory | 0.689 | 0.135 | |
| Aiming training | 0.163 | 0.607 | |
| Stroop | 0.805 | 0.254 | |
| Sequence memory | 0.899 | 0.255 | |
| Hand eye | 0.372 | 0.653 | |
| SS Loadings | 2.134 | 1.350 | |
| Proportion var | 0.356 | 0.225 | |
| Cumulative var | 0.356 | 0.581 |
KMO: Kaiser–Meyer–Olkin index.
The composite scores for the two factors were calculated using the factor score coefficients obtained from the EFA. The scores of the two dimensions were then plotted as boxplots to illustrate their distributions across the four WBGT conditions (Figure 8b). The figure reaffirms the distinct patterns: performance in the sensorimotor reactivity dimension rises steadily with increasing temperature, whereas performance in executive-memory dimension peaks at moderate temperatures and then declines, forming an inverted-U curve. These trends highlight the varying thermal sensitivity of different cognitive types.
4.1.3 The relationship between thermal comfort and cognitive performance
To further explore how subjective thermal comfort (TSV, TPV, and TCV) relates to cognitive performance across the two dimensions (Figure 9), contour plots were generated. The contour gradients range from blue (indicating lower cognitive performance) to red (indicating higher cognitive performance), providing a visual representation of performance variation across different subjective thermal comfort states. Gray regions in the plots indicate areas outside the convex hull of the observed data. As shown in Figure 9a, in the TSV–TPV space, the optimal performance region (dark red) for the executive-memory dimension is confined to the area where TSV ranges from 0 to +1 and TPV equals +1. Similarly, in the TSV–TCV space, the highest performance occurs when TSV is between 0 and +1 and TCV ranges from +1 to +2. The lowest performance zones (deep blue) are predominantly located in the corners of both contour plots, where thermal conditions diverge significantly from thermal neutrality and subjective comfort. In contrast, in Figure 9b, the zone of optimal performance for simple reaction tasks is shifted slightly toward the lower-right regions of the plots. This high-performance zone lies approximately within TSV from +2 to +3, TPV from -2 to -1, and TCV from -2 to 0.
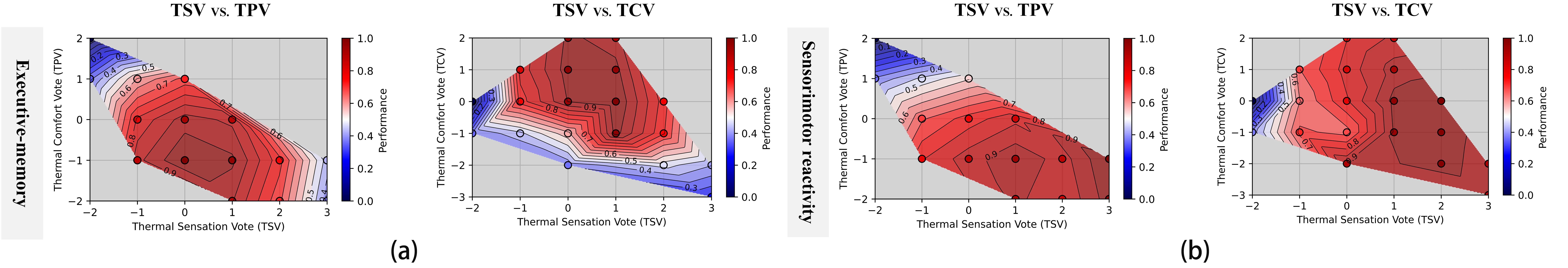
Figure 9. The contour plots of thermal comfort and cognitive performance (a) Thermal comfort and cognitive performance in complex cognitive tasks; (b) Thermal comfort and cognitive performance in simple reaction tasks. TSV: thermal sensation vote; TPV: thermal preference vote; TCV: thermal comfort vote.
These results suggest that performance in the executive- memory dimension is more dependent on thermal comfort. Participants achieved their best results under conditions near thermoneutrality with moderate thermal preference and comfort ratings. In contrast, performance in the sensorimotor reactivity dimension appeared less sensitive to variations in thermal comfort. Participants were able to maintain or even enhance performance under thermally suboptimal conditions.
4.2 EEG relative band power analysis
4.2.1 Global trends in band power under different four temperatures
As shown in Figure 10a, the relative power distributions of the four EEG frequency bands are illustrated across four WBGT conditions by boxplot. Each boxplot is overlaid with a dashed line indicating the trend in mean relative power
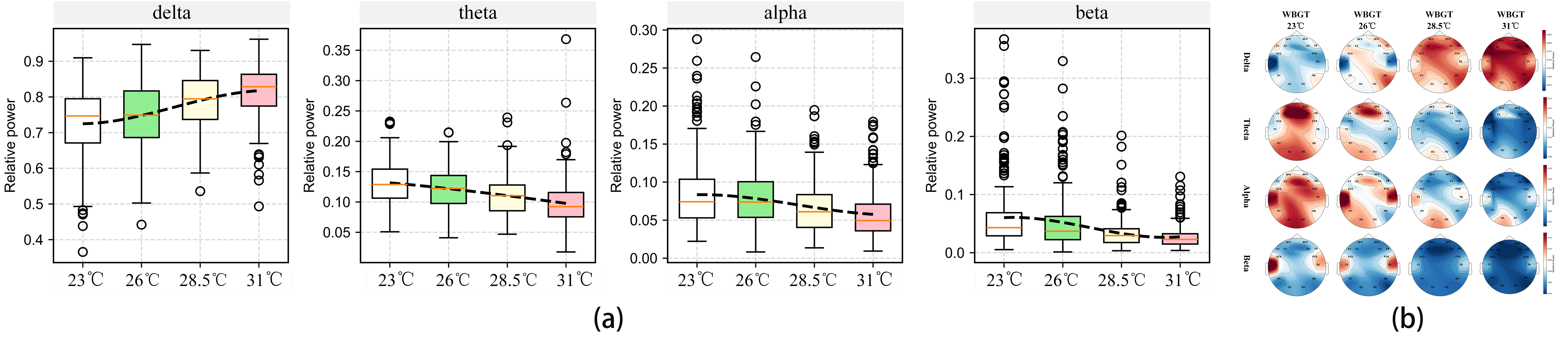
Figure 10. EEG relative band power analysis (a) Global trends in band power; (b) Scalp topographies under different temperatures. EEG: electroencephalogram.
4.2.2 Scalp topographies band power under different temperatures
Figure 10b illustrates the scalp topographies of relative EEG power for each frequency band under different WBGT levels. In the δ band, power localizes predominantly to frontal and occipital cortices. Frontal δ activity exhibits a gradual increase as WBGT rises, whereas occipital δ power demonstrates an initial augmentation followed by a subsequent decrease at the highest thermal load. The θ band likewise engages both frontal and occipital regions, yet shows a continuous attenuation in power with progressive heat stress. α-band oscillations are observed across frontal, temporal, and occipital sites. Frontal and temporal α power declines observably with increasing WBGT, while occipital α undergoes an early suppression before partial recovery at the highest thermal load. Finally, β-band activity remains confined to the temporal lobes and diminishes progressively across all examined thermal conditions.
4.3 Correlations analysis between EEG band power and cognitive performance
To neuroscientifically elucidate how heat stress differentially affects cognitive performance, this study further examined the relationship between EEG frequency-band power (δ, θ, α, and β) and performance across both cognitive dimensions. It specifically compared their overall relationship curves and analyzed differences in EEG-cognition coupling patterns.
4.3.1 Overall tendency between cognitive performance and EEG band power
Figure 11 presents eight scatterplots (4 EEG bands × 2 dimensions) of relative band power (x-axis) versus performance score (y-axis). Each scatterplot is overlaid with a locally weighted scatterplot smoothing (LOWESS)[65] curve and its 95% confidence band (pink ribbon).
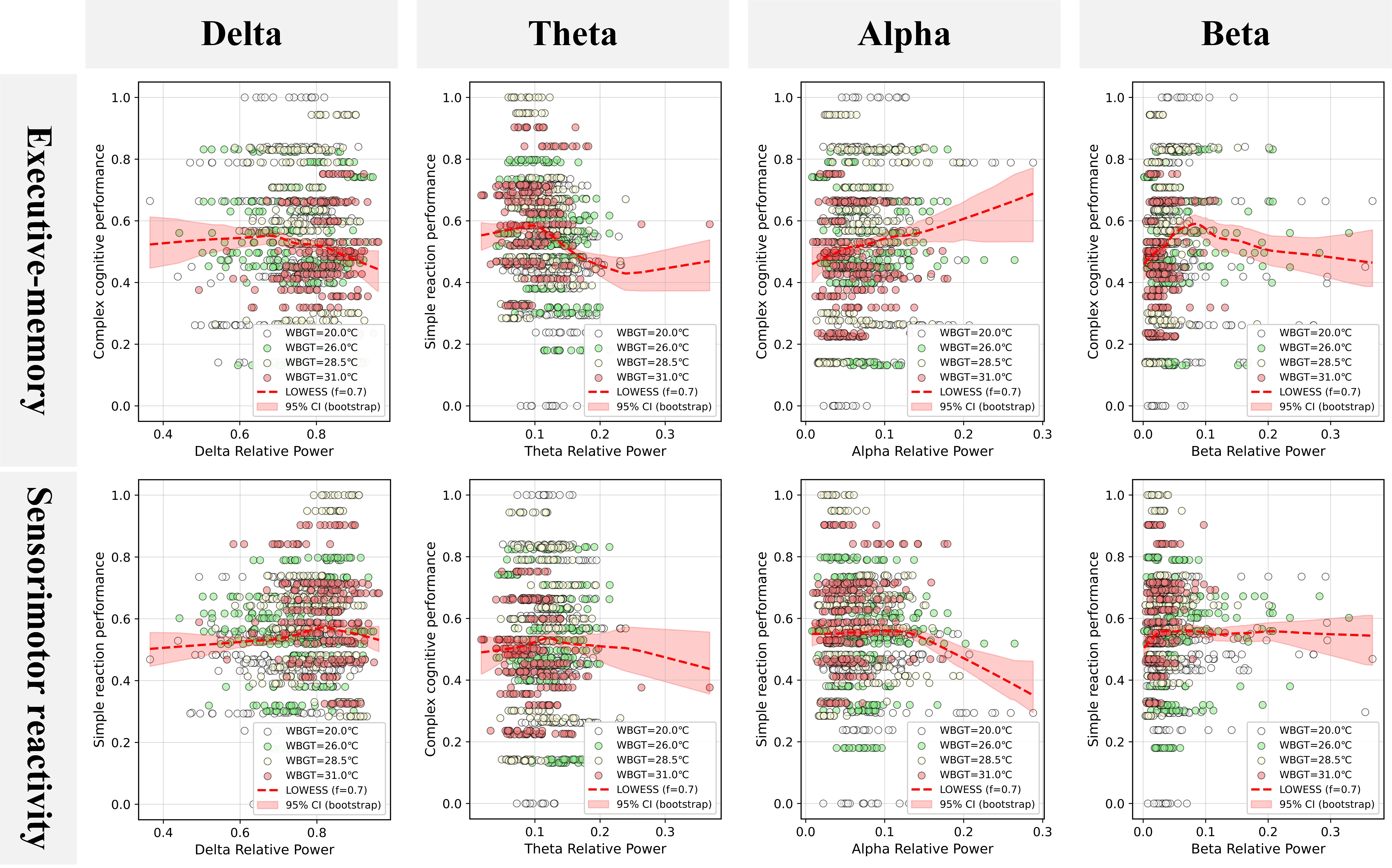
Figure 11. Relationship between EEG band-power and cognitive performance. EEG: electroencephalogram.
The LOWESS curves reveal both similar and distinct band-specific patterns in neural activity across the two dimensions of cognitive performance. In the δ band, the performance of both dimensions peak mid-range, but in the executive-memory dimension, it exhibits a steep rise and fall with a narrow plateau near
To sum up, while both dimensions of cognitive performance generally follow similar overarching trends in, the inverted U pattern is often more pronounced in the executive-memory dimension, and the specific “turning points” differ between two dimensions.
4.3.2 Spatial coupling between EEG band power and cognitive performance
To localize the scalp regions where EEG power sensitively reflects cognitive performance, the maximal information coefficient (MIC) was computed for each of the 14 electrodes. MIC, introduced by Reshef et al.[66], quantifies the strength of both linear and non-linear associations by finding the maximal normalized mutual information over all grid partitions of the scatterplot. The formula for MIC calculation is shown in Equation (6). MIC lies between 0 and 1. A value near 1 indicates a strong functional or arbitrary structure in the data, while a value near 0 denotes no dependency.
Where,
This procedure yielded 14 MIC coefficients per band, one for each electrode. These values were then displayed both as a matrix plot and as an interpolated scalp topography (Figure 12a). Using this spatial mapping, the electrodes exhibiting the highest MIC coefficients were identified, thereby pinpointing the scalp regions where cognitive performance and EEG band power are likely coupled. Additionally, to capture the overall heterogeneity, or “complexity”, of the coupling, Shannon entropy[67] was computed for each task domain (Equations 7 and 8).
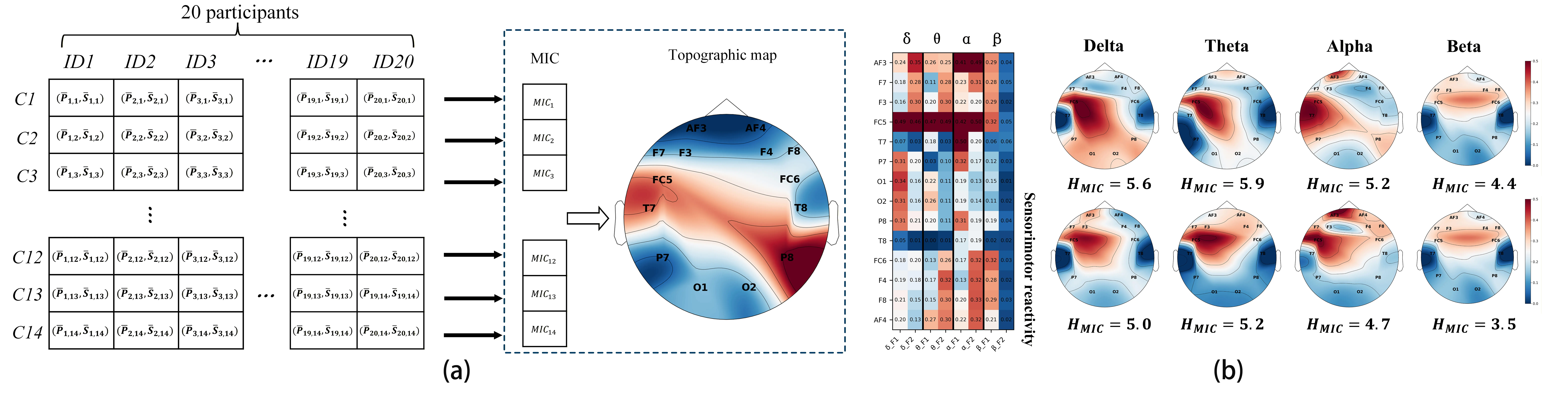
Figure 12. The topographic coupling map of EEG band power and cognitive performance. (a) The approach for topographic coupling in EEG-cognition mapping; (b) Spatial distribution and entropy of EEG–performance coupling for executive-memory and sensorimotor tasks. EEG: electroencephalogram.
Where, the Shannon entropy (HMIC) quantifies the degree to which EEG–behavior coupling is dispersed versus focused across the scalp. A low entropy (near 0) indicates that coupling is highly focal, dominated by one or a few electrodes, whereas a high entropy reflects a broadly distributed, uniform coupling pattern.
As Figure 12b illustrates, δ, θ, and α bands show stronger EEG–performance coupling in the left frontal lobe for both cognitive dimensions, with AF3 and FC5 exhibiting particularly high coupling strength. However, the executive-memory dimension additionally involved elevated MIC values in temporal and occipital regions. Conversely, in the sensorimotor reactivity dimension, posterior coupling was markedly attenuated, most notably in the β band. Quantitatively, entropy values from 14-channel MIC distributions confirmed this spatial heterogeneity: executive-memory tasks showed significantly higher entropy (δ: 5.6, θ: 5.9, α: 5.2, β: 4.4 bits) for a more widespread coupling, while sensorimotor reactivity displayed lower entropy (δ: 5.0, θ: 5.2, α: 4.7, β: 3.5 bits), implying a more localized pattern.
5. Discussion
5.1 Distinctive cognitive performance under heat stress
One major finding of this study is that workers in heat-exposed manual occupations exhibit distinct cognitive performance under heat stress, depending on cognitive task demands. Specifically, workers’ performance in the executive-memory demanding tasks (involving higher cognitive load) exhibits the classic inverted-U relationship with heat stress. Conversely, their performance on the sensorimotor reactivity demanding tasks (involving lower cognitive load) shows the monotonic increasing trend. Although prior research has noted task-specific differences under heat stress[25,30,37], this study reveals a critical nuance for workers with heat-exposed manual occupations. These results suggest that workers with heat-exposed manual occupations might require, or even benefit from, slightly higher heat loads to achieve optimal cognitive performance, especially on the sensorimotor reactivity dimension. This crucial distinction likely stems from occupational heat acclimatization: years of exposure to challenging work environments appear to have shifted their physiological and psychological baselines.
It is notable that the peak in executive-memory performance occurred at WBGTs of 26 °C and 28.5 °C conditions classified as “Medium” and “High” risk by ISO 7243. Two primary factors may account for this phenomenon. First, given that the participants were manual workers from heat-exposed sectors, they likely possess a degree of thermal adaptation that reduces their sensitivity to non-extreme thermal environments; however, confirming this hypothesis warrants further investigation into individual differences in heat tolerance. Second, for this specific demographic, moderate thermal stimulation appears to align with the optimal performance zones described by the Yerkes-Dodson Law and the Maximal Adaptability Model[16,28,29]. This level of heat stress likely induced optimal physiological arousal to prevent under-stimulation, while simultaneously triggering compensatory psychological mechanisms to actively maintain focus, thereby optimizing executive function.
5.2 Neuroscientific insights into cognitive performance under heat stress
This study confirmed the overall tendency and the sensitive cortex region in EEG band power as heat stress intensified. Intensifying heat raises δ - band power while suppressing α, θ, and β bands, which is consistent with major existing literature[23,25,49,50]. The frontal cortex is most susceptible to these shifts, with brief heat-induced activations also appearing in temporal and occipital areas[39,47]. Thus, changes in brain oscillations may precede and potentially contribute to alterations in cognitive performance. Specifically, increased δ activity and reduced α, θ, and β activity in the left frontal cortex may serve as early neural markers of cognitive deterioration under heat stress. Crucially, this suppression of high-frequency activity in the frontal cortex-a region pivotal for top-down risk assessment-provides a neurophysiological basis for compromised cognitive safety, suggesting that the brain’s capacity to monitor errors and suppress unsafe behaviors is physiologically weakened, even if basic motor functions remain intact. Nevertheless, this interpretation is preliminary, and establishing causality will require larger samples and advanced modeling approaches.
Beyond these general tendencies, this research provides novel neuroscientific insights into how heat stress differentially impacts cognitive performance across two distinct dimensions. While both cognitive performance dimensions generally follow similar overarching inverted-U trends, the specific optimal band power levels for performance differ between these two dimensions. Furthermore, scalp topography coupling maps revealed that the cognitive performance executive-memory dimension exhibited a spatially more diffuse EEG-cognition coupling pattern compared to the sensorimotor reactivity dimension. This was particularly evident in the temporal and occipital regions, especially within the β band. Given that these posterior areas are critical for multisensory integration and complex processing[68-70], this distinct coupling pattern suggests that tasks requiring broader neural engagement may consequently face greater vulnerability under thermal stress[64,68].
In fact, these distinctive neural response patterns reflect the human brain’s strategic redistribution of attentional resources under heat stress[71,72]. This offers a neuroscientific basis for the observed distinctive performance patterns across the cognitive dimensions. As heat stress intensifies and the human body’s thermoregulatory capacity is strained, the central nervous system reduces high-frequency activity, conserving energy for essential functions, a state known as “conservation mode”[48,71,73]. In this mode, the brain reduces energy expenditure on high-frequency activity associated with complex processes, prioritizing essential functions. This mechanism offers a compelling explanation for why workers’ performance in the demanding executive-memory dimension declines, while their performance on the sensorimotor reactivity dimension might even improve under relatively extreme heat stress, as the brain prioritizes basic survival and reactive functions over more complex cognitive processes.
5.4 Practical and implications
This research reveals distinct patterns in how workers respond to heat stress during different cognitive tasks, offering both theoretical insights and practical implications for workplace design, task management, and neuroergonomic monitoring.
5.4.1 Task–environment adaptation
The findings provide valuable insights for adaptive workplace design and task management, underscoring the importance of aligning environmental conditions, worker status, and task requirements to ensure cognitive safety. A WBGT-based task allocation framework offers practical guidance: when WBGT is below 28.5 °C, all tasks can generally be performed without additional interventions, though work efficiency can be further improved by appropriately increasing the environmental temperature; once this threshold is reached or exceeded, high-cognitive-load and heat-sensitive activities (e.g., tower-crane operation, precision fitting, complex machine control) should be relocated to climate-controlled or actively cooled environments, and workers should be provided with appropriate cooling aids, while their physiological and cognitive status is closely monitored to prevent performance-related accidents. Specifically, under heat stress, scheduling should incorporate planned rest and task rotation to prevent cumulative neurocognitive fatigue and error prone lapses. When cooling measures are effective, tasks can be scheduled under routine supervision. When WBGT reaches or exceeds 28.5 °C, a structured rotation protocol is warranted: critical high demand tasks should be shared among multiple workers who rotate on and off duty, while off duty workers either rest or temporarily shift to simpler, repetitive operations to facilitate recovery. For roles dominated by simple, repetitive work, rest periods may be less frequent, provided that basic safety and comfort requirements are met. This task–environment matching approach helps ensure appropriate role assignment, thereby balancing workload, enhancing safety, and maintaining productivity in heat-exposed industries.
5.4.2 EEG-based monitoring and practical implementation
The identification of effective EEG indicators, their correspondence with cognitive load, and the recognition of sensitive cortical regions (notably frontal sites such as FC5/AF3) provide a robust foundation for optimizing EEG device design and refining machine-learning models. By targeting precise electrode locations and streamlining both feature and electrode sets, future systems could enable real-time monitoring and early detection of cognitive fatigue. Embedding these algorithms and miniature EEG sensors into smart helmets or headbands would further extend such monitoring to real-world, heat-exposed occupations, enabling continuous state assessment and facilitating automated recommendations for schedule adjustments or targeted micro-breaks to prevent performance lapses and safety incidents. Furthermore, integrating EEG-based smart helmets with a centralized worker state monitoring platform would allow supervisors to dynamically assess cognitive and thermal status across teams, reallocating personnel or adjusting task assignments in real time to balance workload, enhance safety, and maintain productivity under varying thermal conditions.
5.5 Limitations and future study
5.5.1 Methodological and metric limitations
This study sheds light on the effects of heat stress on cognitive performance, providing neuroscientific evidence to substantiate these observations; however, it is important to acknowledge several constraints. These results delineate broad trends in single-band relative power but do not establish precise EEG thresholds for cognitive impairment, and the findings remain correlational rather than causal. Furthermore, while the utilization of relative band power in this study served to minimize inter-subject variability in absolute EEG amplitude and provided a standardized index for cross-subject comparison, its inherent mathematical constraints must be acknowledged. Specifically, since the relative power calculation requires the sum of all frequency bands to equal 100%, it is subject to a “mathematical coupling” effect. Under this constraint, a significant elevation in the power of certain frequency bands may lead to a proportional decrease in the relative values of other bands, regardless of changes in their actual absolute neurophysiological activity. Consequently, the observed shifts in higher-frequency bands may partially reflect this compensatory mathematical displacement rather than an independent neural suppression.
5.5.2 Environmental and ecological validity
To ensure participant safety, thermal exposure was capped at a WBGT of 31 °C, leaving responses to more extreme heat untested. The use of a WBGT-controlled chamber also limited ecological validity, as it could not reproduce dynamic workplace stressors such as humidity fluctuations, variable radiant heat, or changes in airflow. Lighting conditions were maintained by a fixed light source and could not be further manipulated. In addition, cabin-level spatial uniformity was not quantified using multi-point measurements; therefore, minor local microclimates cannot be fully excluded. In real-world settings, these environmental stressors fluctuate over time and interact with one another, so the strength and even the direction of the observed EEG–behavior associations and single-band power trends may differ. This mismatch may lead to a gap between laboratory observations and real-world applications, thereby reducing the generalizability of the identified patterns for real-time cognitive monitoring.
5.5.3 Sampling and spatial resolution
The modest sample size of 20 participants, restricted to a relatively homogeneous cohort of middle-aged workers (mid-40s) from the construction, manufacturing, and sanitation sectors, and the fact that each participant was tested only once, together with the limited spatial resolution of the 14-channel Emotiv headset, may have further constrained data quality and reduced the generalizability of the findings. This also leaves individualized analyses statistically underpowered. Furthermore, although the frontal cortex is pivotal for executive–memory functions, whether focusing solely on the left frontal cortex is sufficient to represent heat-related modulation remains uncertain and warrants further investigation.
5.5.4 Directions for future research
To build upon these insights and address current limitations, future research should deploy high-resolution, multi-electrode EEG systems to capture clearer and higher-dimensional data, thereby enabling the identification of additional effective EEG features beyond relative band power. In particular, incorporating complementary metrics such as absolute band power, aperiodic-adjusted spectral measures, band ratios, and complexity features (e.g., differential entropy) may help mitigate the limitations of relative-power-only analyses and improve physiological interpretability. Parallel efforts should employ larger, more diverse cohorts, ideally within more fine-grained experimental paradigms or real-world field trials that better reflect dynamic workplace stressors (e.g., humidity and radiant heat fluctuations, changes in illumination levels, sudden airflow changes, and irregular workload patterns), to capture detailed, ecologically valid neural responses and finer spatial patterns of brain activity. Exploring causal pathways through approaches such as dynamic causal modeling (DCM) and structural equation modeling (SEM), and by incorporating physiological mediators, will further strengthen causal inference. In addition, applying advanced machine-learning techniques will be essential for uncovering nonlinear neural–cognition relationships, clarifying the cooperative contributions of different cortical regions, and supporting personalized prediction and mitigation strategies. Together, these enhancements will pave the way for neuro-informed occupational interventions to safeguard human well-being in an increasingly global warming environment.
6. Conclusion
The present study comprehensively integrated subjective thermal comfort metrics, performance across six cognitive tasks, and detailed EEG band-power analyses to elucidate the effects of heat stress on workers with heat-exposed manual occupations and their underlying neural correlates. The principal findings are as follows:
(1) The cognitive performance across six tasks was categorized into two distinct dimensions: the executive-memory dimension, which peaked in the slightly higher thermoneutral range and then declined in the uncomfortable zone; and the sensorimotor reactivity dimension, which steadily improved as temperature increased, demonstrating greater resilience to heat stress.
(2) This study confirmed the overall tendency that environmental heat stress alters EEG band power: as WBGT rose from 20 °C to 31 °C, δ-band power increased while α, θ, and β power declined, exhibiting region - specific variations across cortical areas.
(3) EEG band power and cognitive performance exhibited a non-linear relationship—predominantly an inverted-U pattern and this was more pronounced in the executive-memory dimension than in the sensorimotor reactivity dimension.
(4) The left frontal cortex was particularly sensitive to thermal-cognitive demands. EEG-cognition coupling patterns varied distinctly between the two cognitive dimensions: the executive-memory dimension elicited diffuse scalp coupling, whereas the sensorimotor reactivity dimension produced more focal coupling.
This research transcends a mere observation of heat’s impact on workers; it profoundly illuminates the intricate interplay between environmental thermal stress, human cognition, and neural activity. By unveiling the differential vulnerability of cognitive functions and their distinct neurophysiological signatures under heat stress, this study not only substantiates the invisible cognitive burden on heat-exposed manual workers but also fundamentally redefines the approach to occupational safety through the critical lens of cognitive safety. It underscores the critical need to move beyond simple physiological thresholds, advocating for a neuro-informed paradigm that champions proactive, personalized interventions.
Acknowledgement
The authors would like to thank all participants for their time and cooperation.
Authors contribution
Zhang H, Zhao C: Conceptualization, methodology, investigation, formal analysis, writing-original draft.
Li H: Methodology, formal analysis, writing-review & editing.
Hou H: Supervision, funding acquisition, formal analysis, writing-review & editing.
Chong D: Formal analysis, writing-review & editing.
Lan H: Methodology, formal analysis, writing-review & editing.
Conflicts of interest
The authors declare no conflicts of interest.
Ethical approval
The entire experimental protocol was approved by the Ethics Committee of the Hong Kong Polytechnic University (HSEARS20250623002).
Consent to participate
All participants received a detailed briefing on the study protocol and provided written informed consent prior to participation.
Consent for publication
Not applicable.
Availability of data and materials
Not applicable.
Funding
This work was supported by the Hong Kong Polytechnic University (Project ID: P0052446).
Copyright
© The Author(s) 2026.
References
-
1. Parker DE. Urban heat island effects on estimates of observed climate change. Wires Clim Change. 2010;1(1):123-133.[DOI]
-
2. Climate Central [Internet]. Climate change and the escalation of global extreme heat. 2025. Available from: https://www.climatecentral.org/report/climate-change-and-the-escalation-of-global-extreme-heat
-
3. Mohammad Harmay NS, Choi M. The urban heat island and thermal heat stress correlate with climate dynamics and energy budget variations in multiple urban environments. Sustain Cities Soc. 2023;91:104422.[DOI]
-
4. Hong C, Wang Y, Gu Z. How to understand the heat island effects in high-rise compact urban canopy? City Built Enviro. 2023;1:2.[DOI]
-
5. Song J, Chen W, Zhang J, Huang K, Hou B, Prishchepov AV. Effects of building density on land surface temperature in China: Spatial patterns and determinants. Landsc Urban Plan. 2020;198:103794.[DOI]
-
7. Cannady R, Warner C, Yoder A, Miller J, Crosby K, Elswick D, et al. The implications of real-time and wearable technology use for occupational heat stress: A scoping review. Saf Sci. 2024;177:106600.[DOI]
-
9. Nath A, Sahu S, Lee JKW. An umbrella review of systematic reviews and meta-analyses on occupational heat exposure, health risks, and productivity losses globally. Curr Environ Health Rep. 2026;13(1):2.[DOI]
-
11. Vitti M, Padovano A, Facchini F. A review on cognitive workload for industry 5.0. Comput Ind Eng. 2025;207:111350.[DOI]
-
12. Pooladvand S, Hasanzadeh S. Impacts of stress on workers’ risk-taking behaviors: Cognitive tunneling and impaired selective attention. J Constr Eng Manage. 2023;149(8):04023060.[DOI]
-
13. Schmit C, Hausswirth C, Le Meur Y, Duffield R. Cognitive functioning and heat strain: Performance responses and protective strategies. Phys Med. 2017;47(7):1289-1302.[DOI]
-
14. Cramer MN, Gagnon D, Laitano O, Crandall CG. Human temperature regulation under heat stress in health, disease, and injury. Physiol Rev. 2022;102(4):1907-1989.[DOI]
-
15. Reitmayer A, Koth SC, Kobas B, Johnstone KR, Cook MM, Madigan C, et al. Effects of dynamic thermal conditioning on cognitive load and performance in an office environment. Appl Ergon. 2025;122:104395.[DOI]
-
17. Brimicombe C, Gao C, Otto IM. Vulnerable to heat stress: Gaps in international standard metric thresholds. Int J Biometeorol. 2024;68(12):2495-2506.[DOI]
-
18. Liu W, Tian X, Tao M. A model to quantify the relation between cognitive performance and thermal responses in high temperature at a moderate activity level. Build Environ. 2022;207:108431.[DOI]
-
19. Tyner FS, Knott JR. Fundamentals of EEG technology: basic concepts and methods. 1st ed. Philadelphia (PA): Lippincott Williams & Wilkins; 1983. Avaialable from: https://books.google.com/books?hl=en&lr=&id=KDL0E2OtUvAC&oi=fnd&pg=PA3
-
20. Li J, Wu W, Jin Y, Zhao R, Bian W. Research on environmental comfort and cognitive performance based on EEG+VR+LEC evaluation method in underground space. Build Environ. 2021;198:107886.[DOI]
-
21. Cudeck R. Exploratory factor analysis. In: Tinsley HEA, Brown SD, editors. Handbook of Applied Multivariate Statistics and Mathematical Modeling. San Diego: Academic Press; 2000. p. 265-296.[DOI]
-
22. Ren R, Lin D, Zhou S, Zhen M, Sheng Z. Study on the relationship between human EEG and comfort in indoor office spaces under thermal-acoustic interaction environments. Measurement. 2026;257:118961.[DOI]
-
23. Lang X, Wang Z, Tian X, Wu Y, Zhu S, Liu W. The effects of extreme high indoor temperature on EEG during a low intensity activity. Build Environ. 2022;219:109225.[DOI]
-
24. Choi Y, Kim M, Chun C. Effect of temperature on attention ability based on electroencephalogram measurements. Build Environ. 2019;147:299-304.[DOI]
-
25. Niu H, Zhai Y, Huang Y, Wang X, Wang X. Investigating the short-term cognitive abilities under local strong thermal radiation through EEG measurement. Build Environ. 2022;224:109567.[DOI]
-
26. U.S. Bureau of Labor Statistics [Internet]. Employed persons by detailed industry and age. 2024. Available from: HYPERLINK "https://www.bls.gov/cps/cpsaat18b.htm" https://www.bls.gov/cps/cpsaat18b.htm
-
27. Yeoman K, Weakley A, DuBose W, Honn K, McMurry T, Eiter B, et al. Effects of heat strain on cognitive function among a sample of miners. Appl Ergon. 2022;102:103743.[DOI]
-
28. Yerkes R, Dodson J. The relation of strength of stimulus to rapidity of habit-formation. Journal of comparative neurology and psychology. 1908;18(5):459-482. Available from: HYPERLINK "https://psychclassics.yorku.ca/Yerkes/Law/" https://psychclassics.yorku.ca/Yerkes/Law/
-
30. Grether W. Human performance at elevated environmental temperatures. Aerosp Med. 1973;44(7):747-755. Available from: https://asma.kglmeridian.com/view/journals/aeme/44/7/article-p747.xml
-
32. Ramsey JD, Kwon YG. Recommended alert limits for perceptual motor loss in hot environments. Int J Ind Ergon. 1992;9(3):245-257.[DOI]
-
33. Frescura A, Lee PJ, Jeong JH, Soeta Y. Electroencephalogram (EEG) responses to indoor sound sources in wooden residential buildings. Inter Noise. 2021;263(4):1989-1998.[DOI]
-
34. Nayak T, Zhang T, Mao Z, Xu X, Zhang L, Pack DJ, et al. Prediction of human performance using electroencephalography under different indoor room temperatures. Brain Sci. 2018;8(4):74.[DOI]
-
35. Kim H, Hong T, Kim J, Yeom S. A psychophysiological effect of indoor thermal condition on college students’ learning performance through EEG measurement. Build Environ. 2020;184:107223.[DOI]
-
36. Coelli S, Calcagno A, Cassani CM, Temporiti F, Reali P, Gatti R, et al. Selecting methods for a modular EEG pre-processing pipeline: An objective comparison. Biomed Signal Process Control. 2024;90:105830.[DOI]
-
37. Li Y, Li S, Gao W, Xu W, Xu Y, Wang J. Exploring the effects of indoor temperature on college students’ physiological responses, cognitive performance and a concentration index derived from EEG signals. Dev Built Environ. 2022;12:100095.[DOI]
-
38. Zheng H, Pan L, Li T. Research on indoor thermal sensation variation and cross-subject recognition based on electroencephalogram signals. J Build Eng. 2023;76:107305.[DOI]
-
39. Libenson M. Practical approach to electroencephalography E-Book [Internet]. Philadelphia: Elsevier Health Sciences; 2024. Available from: https://books.google.com/books?hl=en&lr=&id=Ck_9EAAAQBAJ&oi=fnd&pg=PP1
-
41. Beste C, Münchau A, Frings C. Towards a systematization of brain oscillatory activity in actions. Commun Biol. 2023;6:137.[DOI]
-
42. Lu M, Hu S, Mao Z, Liang P, Xin S, Guan H. Research on work efficiency and light comfort based on EEG evaluation method. Build Environ. 2020;183:107122.[DOI]
-
43. Lan L, Lian Z. Use of neurobehavioral tests to evaluate the effects of indoor environment quality on productivity. Build Environ. 2009;44(11):2208-2217.[DOI]
-
44. Apicella A, Isgrò F, Pollastro A, Prevete R. On the effects of data normalization for domain adaptation on EEG data. Eng Appl Artif Intell. 2023;123:106205.[DOI]
-
45. Akbulut O. Feature normalization effect in emotion classification based on EEG signals. Sakarya Univ J Sci. 2020;24(1):60-66.[DOI]
-
46. Erkan İ. Cognitive response and how it is affected by changes in temperature. Build Res Inf. 2021;49(4):399-416.[DOI]
-
47. Li S, Li Y, Gao W, Xiao F. A comprehensive study on the impacts of thermal comfort on occupants’ thermophysiology and cognitive performances in a radiant cooling environment using physiological measurements. Energy Build. 2024;323:114771.[DOI]
-
48. Shin Y, Ham J, Cho H. Experimental study of thermal comfort based on driver physiological signals in cooling mode under summer conditions. Appl Sci. 2021;11(2):845.[DOI]
-
49. Mansi SA, Pigliautile I, Porcaro C, Pisello AL, Arnesano M. Application of wearable EEG sensors for indoor thermal comfort measurements. Acta Imeko. 2021;10(4):214.[DOI]
-
50. Jianhui S, Li T, Songtao H. Human thermal comfort under temperature step-change environment based on electroencephalography. Sci Technol Eng. 2022;22(21):9054-9060. Available from: https://www.stae.com.cn/jsygc/article/html/2109930
-
51. Run K, Cévaër F, Dubé JF. Influence of transient conditions on thermal comfort evaluations: Field study in university buildings. Case Stud Therm Eng. 2025;66:105737.[DOI]
-
52. Bai H, Lu Y, Cao X, Zhuo S. The practical impact of indoor temperature on the productivity of prefabricated construction workers using electroencephalogram data. Sci Rep. 2025;15:28670.[DOI]
-
54. Tao M, Yang D, Liu W. Learning effect and its prediction for cognitive tests used in studies on indoor environmental quality. Energy Build. 2019;197:87-98.[DOI]
-
55. Human Benchmark [Internet]. Cognitive tests. 2026. Available from: https://humanbenchmark.com/
-
56. Bendak S, Jouaret R, Rashid H. Effects of high ambient temperature on construction workers performance: A longitudinal empirical study. J Saf Res. 2022;81:197-202.[DOI]
-
62. Hsieh JC, Alawieh H, Li Y, Iwane F, Zhao L, Anderson R, et al. A highly stable electrode with low electrode-skin impedance for wearable brain-computer interface. Biosens Bioelectron. 2022;218:114756.[DOI]
-
63. Dimitrakopoulos G. From raw EEG signals to brain networks: An EEGLAB tutorial. In: Vlamos P, Kotsireas IS, Tarnanas I, editors. Handbook of Computational Neurodegeneration. Cham: Springer; 2023. p. 1-16.[DOI]
-
64. Zhang N, Liu C, Li J, Hou K, Shi J, Gao W. A comprehensive review of research on indoor cognitive performance using electroencephalogram technology. Build Environ. 2024;257:111555.[DOI]
-
65. Cleveland WS. Robust locally weighted regression and smoothing scatterplots. J Am Stat Assoc. 1979;74(368):829-836.[DOI]
-
66. Reshef DN, Reshef YA, Finucane HK, Grossman SR, McVean G, Turnbaugh PJ, et al. Detecting novel associations in large data sets. Science. 2011;334(6062):1518-1524.[DOI]
-
67. Lin J. Divergence measures based on the Shannon entropy. IEEE Trans Inform Theory. 1991;37(1):145-151.[DOI]
-
68. Pascucci D, Menétrey MQ, Passarotto E, Luo J, Paramento M, Rubega M. EEG brain waves and alpha rhythms: Past, current and future direction. Neurosci Biobehav Rev. 2025;176:106288.[DOI]
-
69. Michail G, Senkowski D, Holtkamp M, Wächter B, Keil J. Early beta oscillations in multisensory association areas underlie crossmodal performance enhancement. NeuroImage. 2022;257:119307.[DOI]
-
70. Lundqvist M, Miller EK, Nordmark J, Liljefors J, Herman P. Beta: Bursts of cognition. Trends Cogn Sci. 2024;28(7):662-676.[DOI]
-
72. Zhao W, Su K, Zhu H, Kaiser M, Fan M, Zou Y, et al. Activity flow under the manipulation of cognitive load and training. NeuroImage. 2024;297:120761.[DOI]
Copyright
© The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Publisher’s Note
Share And Cite