Table of Contents
From algorithm to application: AI-powered design of ionizable lipids for mRNA delivery
Artificial intelligence (AI) is revolutionizing the design of ionizable lipids, the pivotal components of lipid nanoparticles (LNPs) for messenger RNA (mRNA) delivery, enabling efficient exploration of vast chemical space of ionizable lipids beyond the ...
More.Artificial intelligence (AI) is revolutionizing the design of ionizable lipids, the pivotal components of lipid nanoparticles (LNPs) for messenger RNA (mRNA) delivery, enabling efficient exploration of vast chemical space of ionizable lipids beyond the reach of traditional methods. This mini-review explores the burgeoning field of AI-powered design and optimization of ionizable lipids for mRNA delivery. We also discuss the critical role of high-throughput experimental strategies, particularly barcoding coupled with next-generation sequencing, in generating the large-scale in vivo datasets for model training. Finally, we discuss current challenges, including data quality and the necessity for domain-specific modeling strategies, and present a future outlook on the integration of AI with scientific computing for LNP research.
Less.Danhong Liang, ... Bo Ying
DOI:https://doi.org/10.70401/bmeh.2026.0026 - April 20, 2026
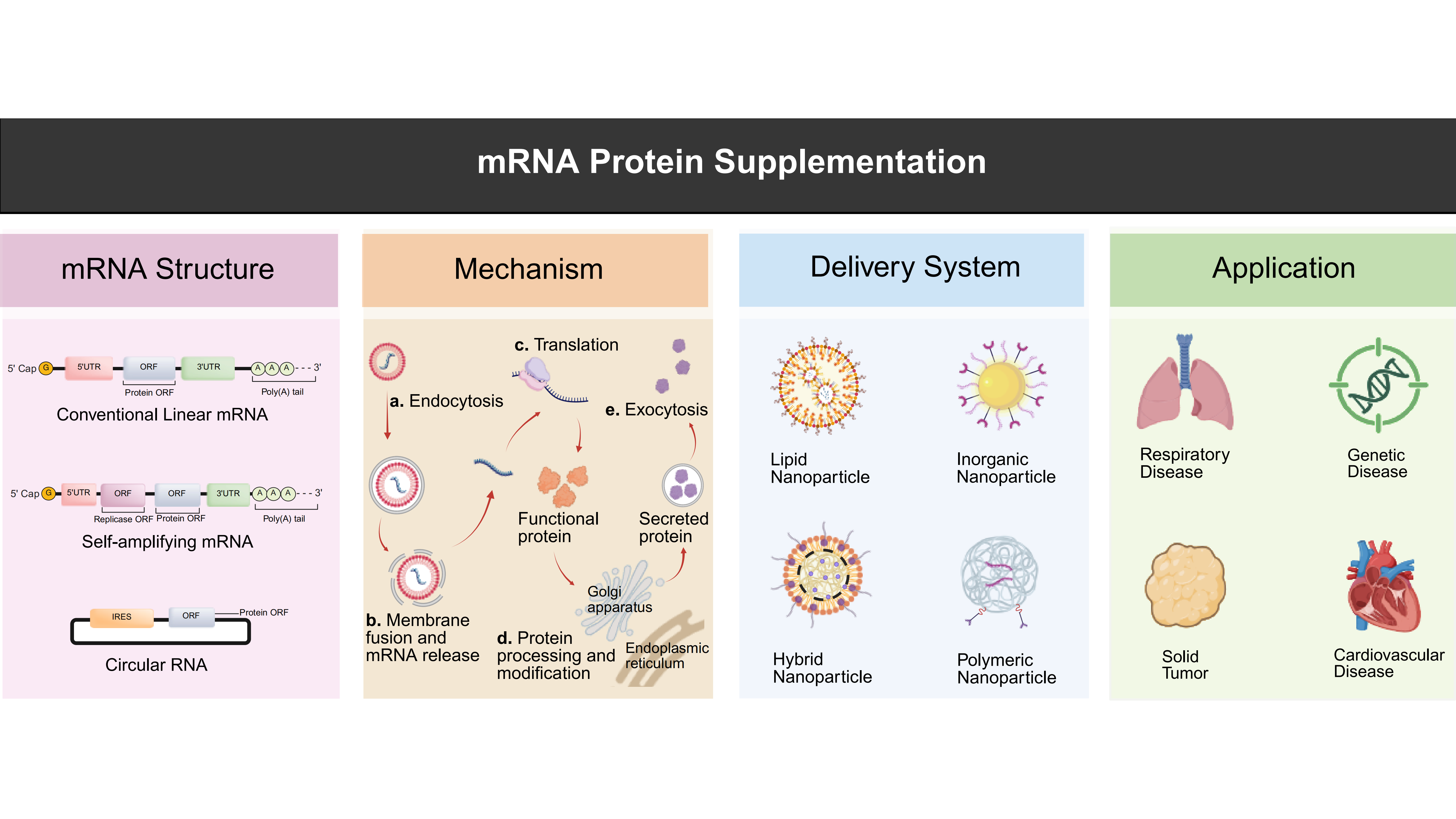
Engineering mRNA therapeutics for protein supplementation: Challenges and future horizons
Messenger RNA (mRNA) protein replacement therapy harnesses synthetic mRNA to direct endogenous protein synthesis, offering a versatile approach to restore or substitute proteins that are absent or dysfunctional in disease. Here, we review recent advances ...
More.Messenger RNA (mRNA) protein replacement therapy harnesses synthetic mRNA to direct endogenous protein synthesis, offering a versatile approach to restore or substitute proteins that are absent or dysfunctional in disease. Here, we review recent advances that have transformed this concept into a promising therapeutic platform, summarizing progress in mRNA design, delivery technologies, and preclinical and clinical applications across metabolic, oncological, and cardiovascular disorders. We also examine persistent challenges, including achieving precise tissue targeting, extending expression duration, and balancing immune tolerance with translation efficiency, that define the next frontier for clinical translation. By systematically analyzing these obstacles and evaluating emerging solutions, such as next-generation mRNA architectures, targeted biomaterial platforms, and programmable expression control, the review proposes new conceptual and technological directions for the next phase of mRNA therapeutic development. Collectively, these insights provide a structure for advancing mRNA protein replacement from proof-of-concept studies toward a broadly applicable platform for precision medicine.
Less.Yiyang Tao, ... Congcong Xu
DOI:https://doi.org/10.70401/bmeh.2026.0025 - April 16, 2026
Engineering biomaterials for microwave medicine: Physicochemical mechanisms and emerging biomedical applications
Microwave (MW) medicine has emerged as a distinct interdisciplinary field, predicated on the unique capacity of non-ionizing electromagnetic radiation to penetrate deep-seated tissues and interact efficiently with biological dielectrics for diverse ...
More.Microwave (MW) medicine has emerged as a distinct interdisciplinary field, predicated on the unique capacity of non-ionizing electromagnetic radiation to penetrate deep-seated tissues and interact efficiently with biological dielectrics for diverse therapeutic and diagnostic applications. Despite its clinical establishment in tumor ablation and hemostasis, conventional MW interventions are largely constrained by non-selective macroscopic heating, leaving the intricate potential of non-thermal biophysical modulation underutilized. The integration of engineered biomaterials provides a transformative framework to bridge this gap, enabling the precise modulation of MW-tissue interactions at the micro- and nanoscale. This review systematically elucidates how rational material design via tuning dielectric and magnetic loss, band-gap engineering, and structural polarization expands MW medicine beyond bulk heating toward controlled biological regulation. We discuss mechanisms where biomaterials function as localized energy antennas to sharpen thermal gradients, as MW-dynamic sensitizers to induce reactive oxygen species generation, and as intelligent interfaces to regulate ionic homeostasis. Representative advancements are summarized across antitumor, antibacterial, and anti-inflammatory therapies, alongside innovations in high-fidelity thermoacoustic imaging. Furthermore, emerging frontiers in non-destructive tissue repair and neuromodulation are highlighted. This review critically examines the design principles and translational challenges of MW-based medical technologies by analyzing correlations between physicochemical parameters and specific biological outcomes. It is expected to advance MW medicine from empirically guided thermal interventions toward mechanism-driven, precision-targeted electromagnetic therapeutics.
Less.Rui Song, ... Liewei Wen
DOI:https://doi.org/10.70401/bmeh.2026.0024 - April 16, 2026
Macrophage-targeted mRNA therapeutics: Emerging strategies for cancer immunotherapy
Tumor-associated macrophages (TAMs) are pivotal regulators of the immunosuppressive tumor microenvironment and major contributors to resistance against conventional and immunotherapeutic interventions. Rather than eliminating TAMs, emerging strategies ...
More.Tumor-associated macrophages (TAMs) are pivotal regulators of the immunosuppressive tumor microenvironment and major contributors to resistance against conventional and immunotherapeutic interventions. Rather than eliminating TAMs, emerging strategies aim to functionally reprogram them toward an antitumor phenotype, a therapeutic objective uniquely enabled by the precise, transient, and non-integrating nature of mRNA, which allows reversible modulation without genomic risk. Recent progress in nanocarrier design has improved selective delivery to TAMs through both passive uptake and active targeting, with administration routes tailored to tumor location. Precise immunomodulatory interventions in macrophages, accomplished by mRNA payloads designed to induce pro-inflammatory polarization, enhance phagocytosis, or block immunosuppressive signals, thereby remodel the tumor immune microenvironment and generate synergy with established treatments. Future efforts might concentrate on macrophage heterogeneity, carrier immunogenicity, and scalable formulation development to advance clinical translation, not only in cancer but also in other diseases shaped by dysregulated macrophage function.
Less.Kedan Gu, Sitao Xie
DOI:https://doi.org/10.70401/bmeh.2026.0023 - April 07, 2026