Table of Contents
Iridium(I)-catalyzed enantioselective five- and six-membered C–H silylation of ferrocenes
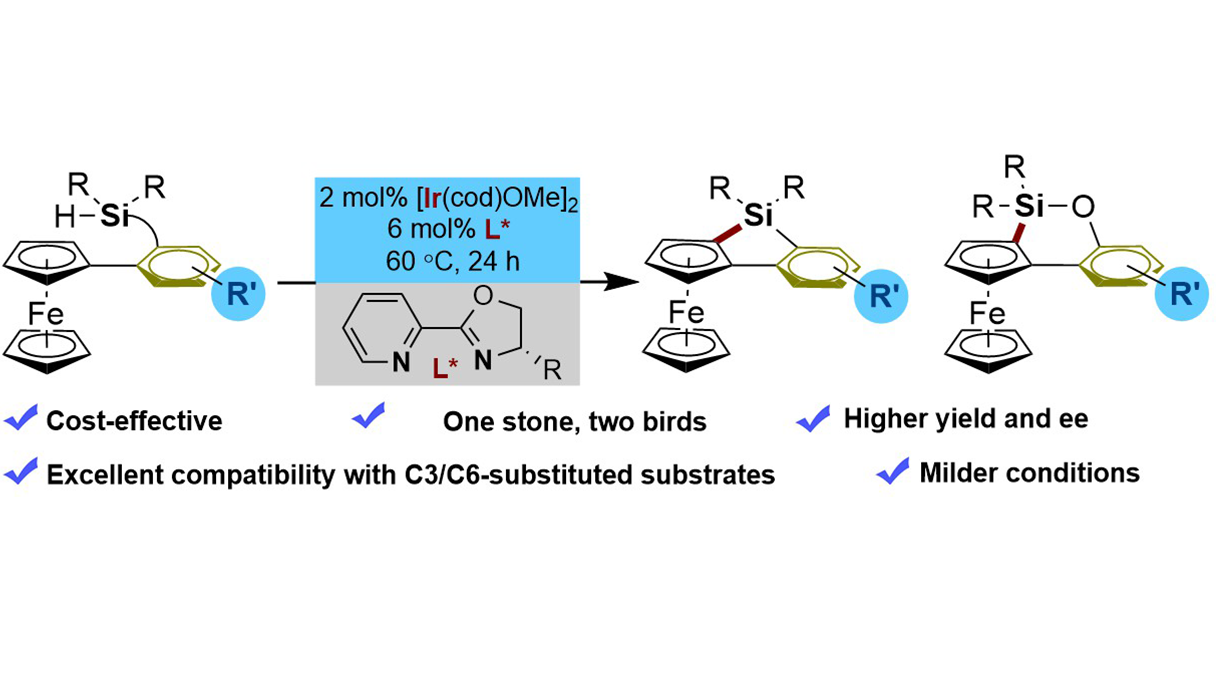
We report the first Ir(I)-catalyzed enantioselective intramolecular C–H silylation of ferrocenes for the efficient construction of planar-chiral 5- and 6-membered ferrocenyl silacycles. This method overcomes key limitations of existing Rh(I)-based ...
More.We report the first Ir(I)-catalyzed enantioselective intramolecular C–H silylation of ferrocenes for the efficient construction of planar-chiral 5- and 6-membered ferrocenyl silacycles. This method overcomes key limitations of existing Rh(I)-based systems, offering superior reactivity, enhanced cost-effectiveness, and broader substrate scope, including challenging C3- and C6-substituted derivatives, as well as excellent enantiocontrol under mild conditions without requiring exogenous H2 acceptors. The utility of the obtained planar-chiral scaffolds is preliminarily demonstrated through the synthesis of novel monophosphine ligands, which exhibit promising activity in asymmetric catalysis.
Less.Fang Gao, ... Dongbing Zhao
DOI:https://doi.org/10.70401/cc.2026.0019 - April 07, 2026
Enantio- and diastereoselective NHC/Cu-catalyzed intermolecular dearomative cyclopropanation of indoles with diazo esters
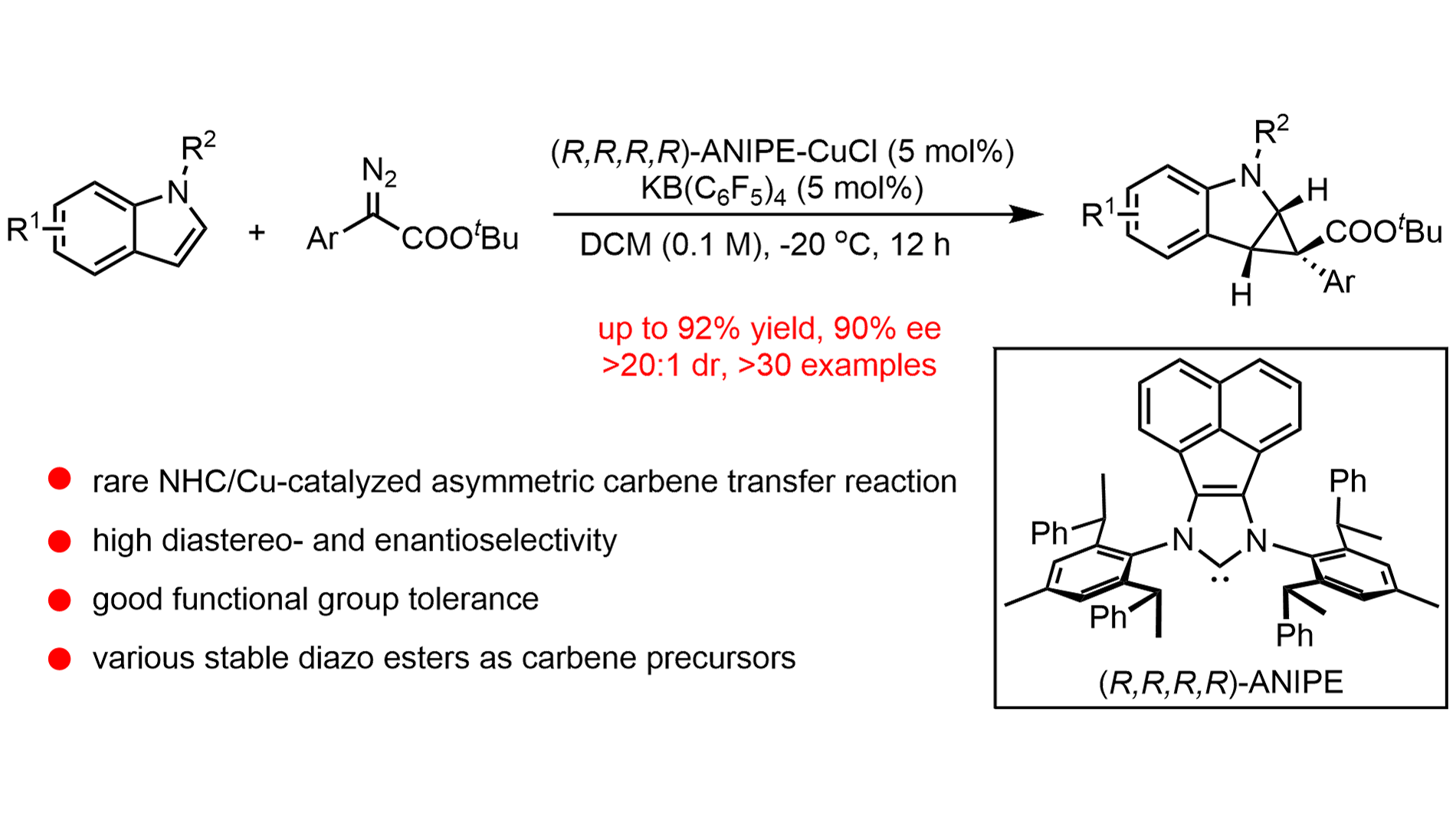
We report a highly enantioselective N-heterocyclic carbene (NHC)/copper-catalyzed intermolecular dearomative cyclopropanation of indoles with diazo esters. This protocol enables the efficient construction of cyclopropane-fused indolines featuring ...
More.We report a highly enantioselective N-heterocyclic carbene (NHC)/copper-catalyzed intermolecular dearomative cyclopropanation of indoles with diazo esters. This protocol enables the efficient construction of cyclopropane-fused indolines featuring quaternary stereogenic centers under mild conditions. A broad range of substituted indoles and diazo esters is tolerated, delivering the corresponding products in high yields with enantioselectivities of up to 90% ee. Notably, the employment of a bulky chiral NHC ligand is crucial for achieving effective stereocontrol over the challenging quasi-linear copper–carbenoid intermediate. Furthermore, gram-scale reactions and downstream transformations demonstrate the synthetic utility of this methodology. Mechanistic investigations, including kinetic isotope effect and Hammett studies, support a reaction pathway involving nucleophilic attack of the indole on a copper–carbenoid species. This earth-abundant copper catalytic system serves as a sustainable alternative to traditional noble-metal-based asymmetric carbene-transfer reactions.
Less.Jiawei Lu, ... Shi-Liang Shi
DOI:https://doi.org/10.70401/cc.2026.0017 - March 24, 2026
Pd(II)-catalyzed atroposelective and enantioselective C−H olefination of [n]metacyclophanes
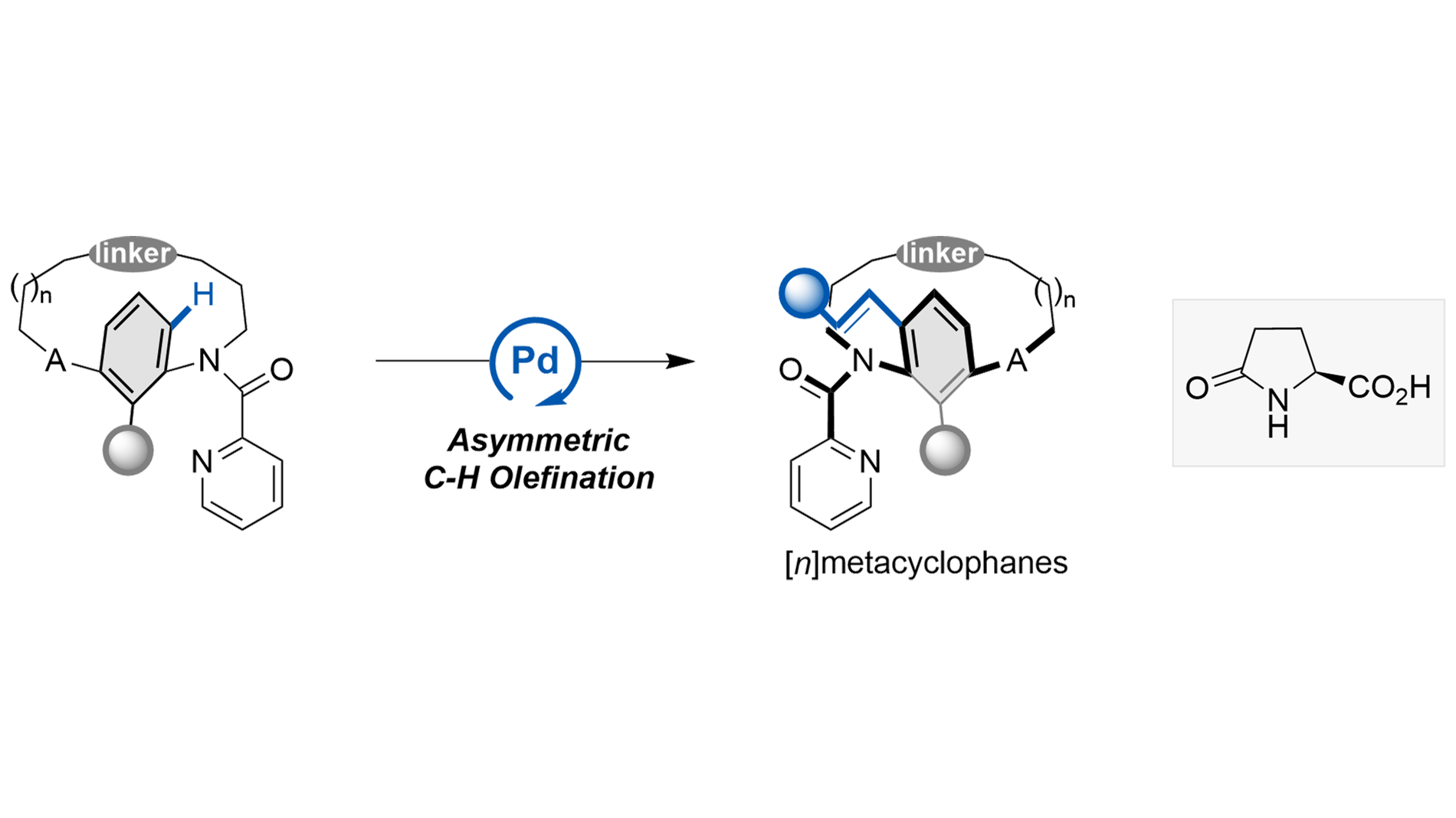
A Pd(II)-catalyzed C–H olefination strategy has been established for the enantioselective synthesis of [n]metacyclophanes. The method demonstrates excellent functional group tolerance across a series of olefins and metacyclophane precursors ...
More.A Pd(II)-catalyzed C–H olefination strategy has been established for the enantioselective synthesis of [n]metacyclophanes. The method demonstrates excellent functional group tolerance across a series of olefins and metacyclophane precursors with varying ansa-chain lengths, achieving high stereocontrol through kinetic or dynamic kinetic resolution. Racemization experiments were conducted to investigate the relationship between molecular structure and conformational stability. Furthermore, the synthetic utility of this approach was demonstrated through gram-scale synthesis and the application of a resulting bifunctional thiourea catalyst in asymmetric Michael additions, highlighting its potential in asymmetric synthesis.
Less.Xin-En Yan, ... Changgui Zhao
DOI:https://doi.org/10.70401/cc.2026.0016 - March 24, 2026
Harnessing molecular motion for asymmetry – nitrogen inversion as a springboard for stereoselective C–H functionalization
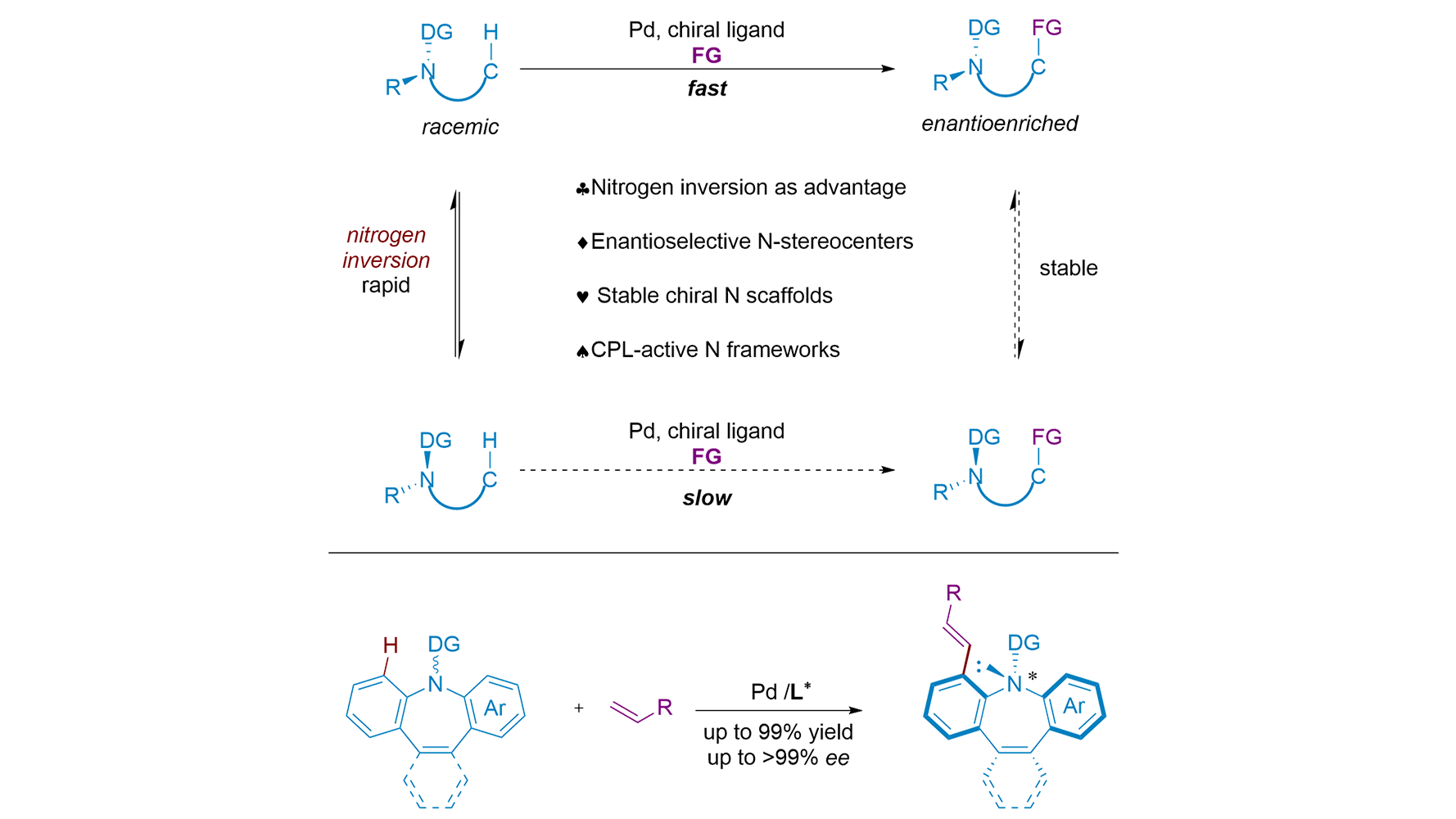
The asymmetric construction of nitrogen stereocenters is notoriously difficult due to rapid nitrogen inversion. Zhang and co-workers now showcase a Pd-catalyzed enantioselective C-H activation strategy that turns this inversion into an advantage, delivering ...
More.The asymmetric construction of nitrogen stereocenters is notoriously difficult due to rapid nitrogen inversion. Zhang and co-workers now showcase a Pd-catalyzed enantioselective C-H activation strategy that turns this inversion into an advantage, delivering stable chiral azepines with high enantioselectivity. Coincidentally, almost at the same time, Shi and co-workers reported a similar C-H alkylation reaction. Beyond methodology, the products serve as promising scaffolds for asymmetric catalysis and chiroptical materials, bridging synthesis and function. This Perspective discusses how their works not only provide a powerful synthetic method but also open a new avenue for constructing heteroatom stereocenters by harnessing, rather than suppressing, molecular dynamics. Here, nitrogen inversion is deliberately exploited as a dynamic feature that enables enantioselective C-H functionalization, rather than being suppressed as a stereochemical liability.
Less.Chun-Yan Guan, Guang-Jian Mei
DOI:https://doi.org/10.70401/cc.2026.0015 - March 11, 2026
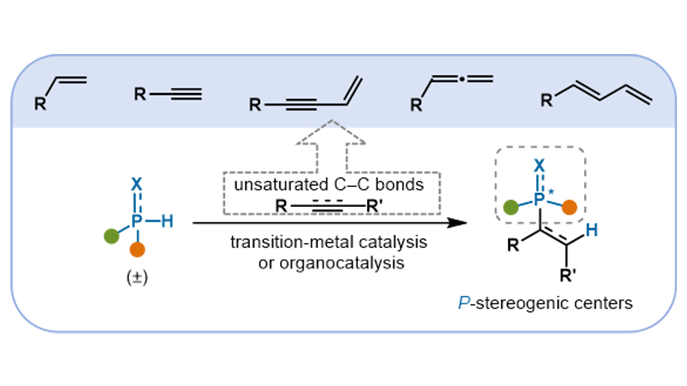
Catalytic construction of P-stereogenic centers through asymmetric hydrophosphination of unsaturated C–C bonds
P-Stereogenic centers are important structural motifs prevalent in natural products, bioactive molecules, and high-performance ligands. Their presence confers significant value across medicinal chemistry, materials science, and asymmetric ...
More.P-Stereogenic centers are important structural motifs prevalent in natural products, bioactive molecules, and high-performance ligands. Their presence confers significant value across medicinal chemistry, materials science, and asymmetric catalysis. In recent years, catalytic asymmetric hydrophosphination has emerged as a powerful and efficient strategy for constructing such P-stereogenic compounds. Distinguished by high atom economy, broad substrate compatibility, and excellent stereocontrol under mild conditions, these transformations align closely with the principles of sustainable and green synthesis. This review summarizes recent advances in the catalytic asymmetric hydrophosphination of unsaturated C–C bonds for the synthesis of P-stereogenic centers. It covers substrates including alkenes bearing electron-withdrawing groups, alkynes, as well as specialized systems such as enynes, allenes, and conjugated dienes. Emphasis is placed on the design of catalytic systems, encompassing transition-metal catalysts (e.g., Pd, Ni, Cu, Co, Mn) and organocatalysts, along with their mechanisms. Current challenges, such as the low reactivity of unactivated or sterically hindered substrates and difficulties in achieving stereochemical differentiation between phosphorus substituents, are also discussed. This review aims to provide a reference for further innovation and methodological development in the synthesis of P-stereogenic molecules.
Less.Bing-Lin Wang, ... Xiao-Hui Yang
DOI:https://doi.org/10.70401/cc.2026.0014 - March 10, 2026
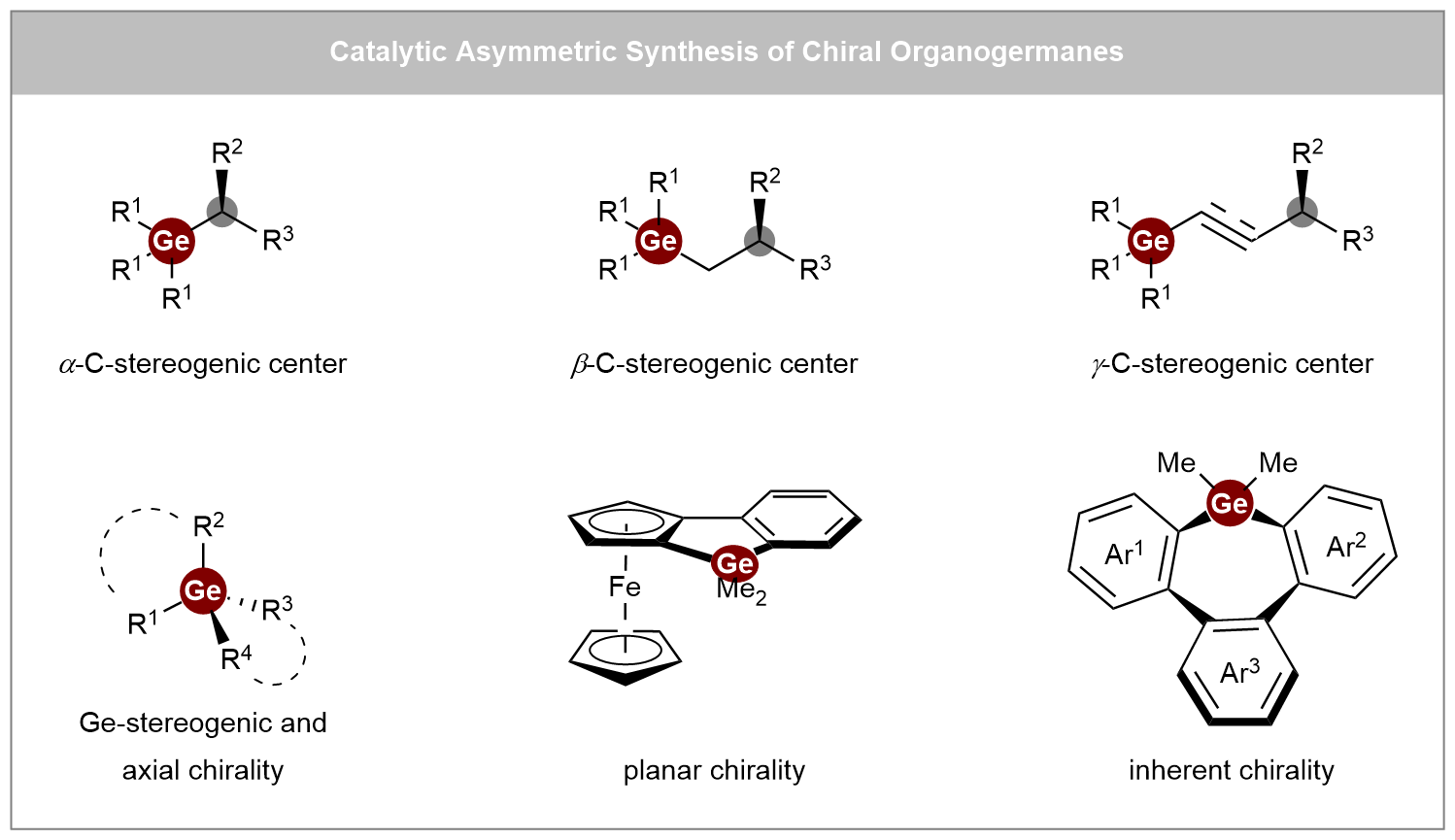
Recent advances in catalytic asymmetric synthesis of chiral organogermanes
Chiral organogermanes hold great potential as bioisosteres in medicinal chemistry and functional materials, yet their development has long been hindered by a scarcity of efficient synthetic strategies. This review offers a comprehensive overview of recent ...
More.Chiral organogermanes hold great potential as bioisosteres in medicinal chemistry and functional materials, yet their development has long been hindered by a scarcity of efficient synthetic strategies. This review offers a comprehensive overview of recent advances in the catalytic asymmetric synthesis of chiral organogermanes, highlighting a shift from traditional resolution methods toward asymmetric catalytic approaches. The content is organized into three main categories: (i) synthesis of C-stereogenic germanes, (ii) synthesis of Ge-stereogenic germanes, and (iii) synthesis of other chiral germanes, including planar, inherent, and axially chiral types. Key synthetic methodologies are systematically examined, such as enantioselective alkene hydrofunctionalization, carbene insertion, coupling reactions, and [2+2+2] cycloaddition, utilizing a variety of catalytic systems ranging from transition metals (Rh, Cu, Ni, Co) and Lewis acids to engineered metalloenzymes. Particular emphasis is placed on the mechanistic insights and ligand design principles that enable stereochemical control in these transformations. We hope this review will inspire chemists working in related areas and contribute to the future advancement of this field.
Less.Shao-Wu Liu, ... Chuan He
DOI:https://doi.org/10.70401/cc.2026.0013 - February 13, 2026