Keyun Ren, CATUG Biotechnology, Suzhou 215000, Jiangsu, China; Wuhan CATUG Biotechnology, Wuhan 430074, Hubei, China. E-mail: keyun.ren@catugbio.com
Abstract
Antibody-targeted lipid nanoparticles (Ab-LNPs) represent a highly promising delivery platform for precision therapy, enabling efficient in vivo targeted delivery of nucleic acid drugs such as mRNA, DNA and siRNA. Currently, the two primary strategies for antibody functionalization of LNPs are the post-insertion method and direct surface conjugation. This review outlines the key chemistry, manufacturing, and controls challenges associated with scaling up of Ab-LNP production, with a focus on antibody modification strategies, process scale-up challenges, and quality control considerations. It aims to provide practical guidance for translating Ab-LNP technology from laboratory research to scalable manufacturing.
Keywords
1. Introduction
Lipid nanoparticles (LNPs) have been firmly established as a robust delivery platform for nucleic acid therapeutics, most notably mRNA, as evidenced by their successful deployment in COVID-19 vaccines[1-3]. However, conventional LNPs are subject to significant hepatic sequestration upon systemic administration, which limits their utility in treating diseases of extrahepatic tissues[4]. To address this constraint and enable active tissue-specific delivery, antibody-targeted lipid nanoparticles (Ab-LNPs) have been developed[5-7]. Through the conjugation of specific antibodies onto the LNP surface, Ab-LNPs acquire the capacity to actively recognize and bind to antigens on target cells, enabling efficient and precise in vivo delivery of nucleic acid drugs. This targeting strategy shows substantial promise across multiple therapeutic areas, including oncology and in vivo cell engineering.
The two predominant methods for antibody functionalization of LNPs are post-insertion and direct surface conjugation (Scheme 1). Post-insertion entails the integration of antibody-lipid conjugates into pre-formed LNPs via membrane fusion, while direct surface conjugation relies on the formation of stable chemical bonds between antibodies and functional groups on the particle surface[8,9]. Both techniques preserve antibody binding activity and present distinct advantages and drawbacks in terms of chemistry, manufacturing, and controls (CMC) during scale-up. A pivotal challenge lies in the judicious selection of a conjugation strategy that ensures manufacturing robustness and batch-to-batch reproducibility.
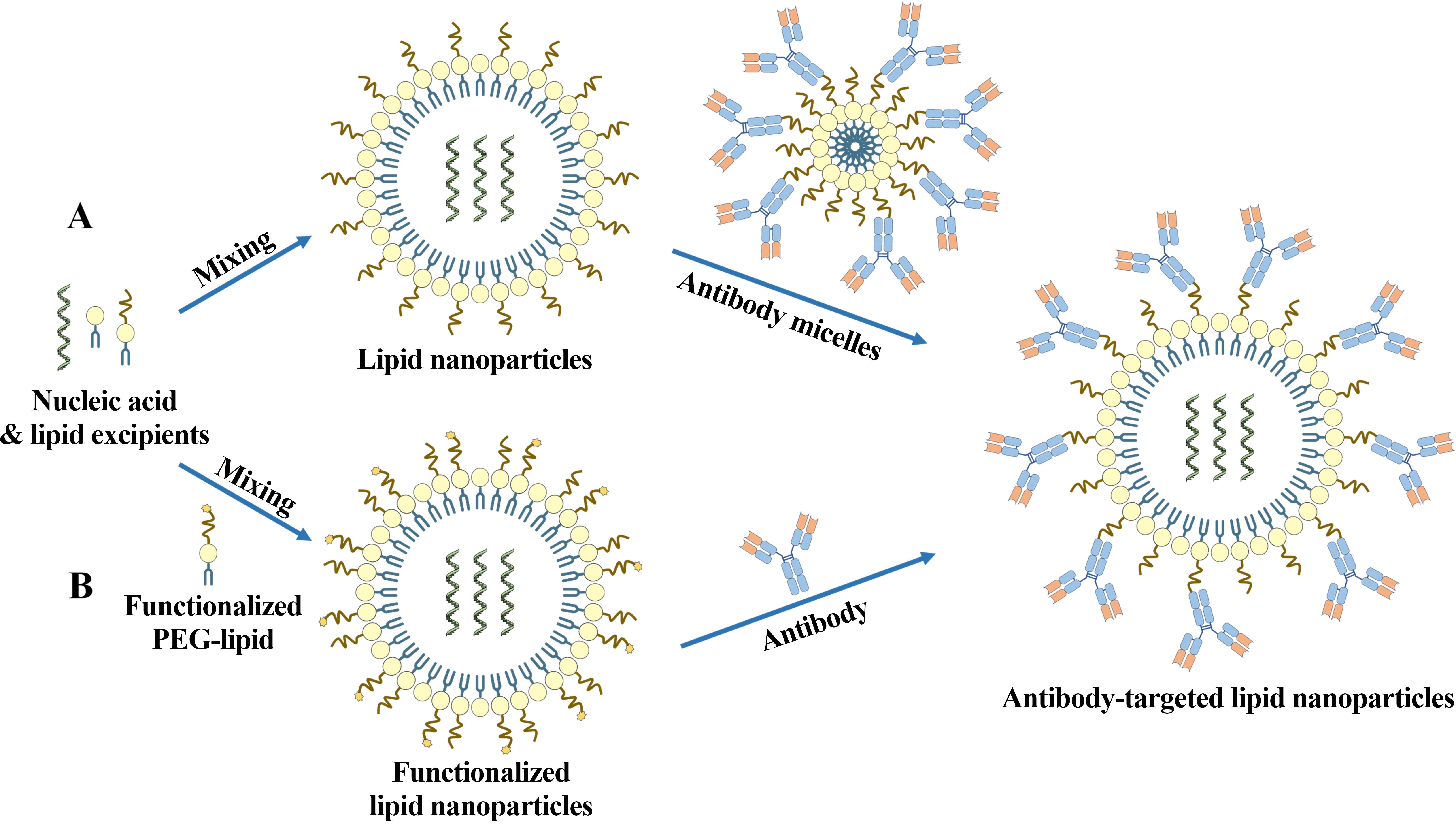
Scheme 1. The two predominant methods for antibody functionalization of LNPs. (A) Post-insertion; (B) Direct surface conjugation. PEG: polyethylene glycol.
Despite promising preclinical results and the progression of several candidates into clinical trials, such as Capstan’s CPTX2309 for
2. Post-Insertion and Direct Surface Conjugation Strategies
Currently, the primary modification methods for Ab-LNPs are post-insertion and direct surface conjugation[12]. Taking thiol-based chemical conjugation as an example, the post-insertion method modularizes the entire process. As shown in Figure 1A, LNP preparation and conjugation are separated into two modules: antibodies are first reduced and then conjugated with maleimide (Mal) groups on the surface of micelles to form antibody-micelles. These micelles are subsequently fused with mRNA-loaded LNPs via thermodynamically-driven hydrophobic interactions, inserting into the lipid membrane surface to obtain Ab-LNPs[13,14]. In contrast, direct surface conjugation involves mixing maleimide lipids (functionalized polyethylene glycol (PEG)-lipids) with other lipids
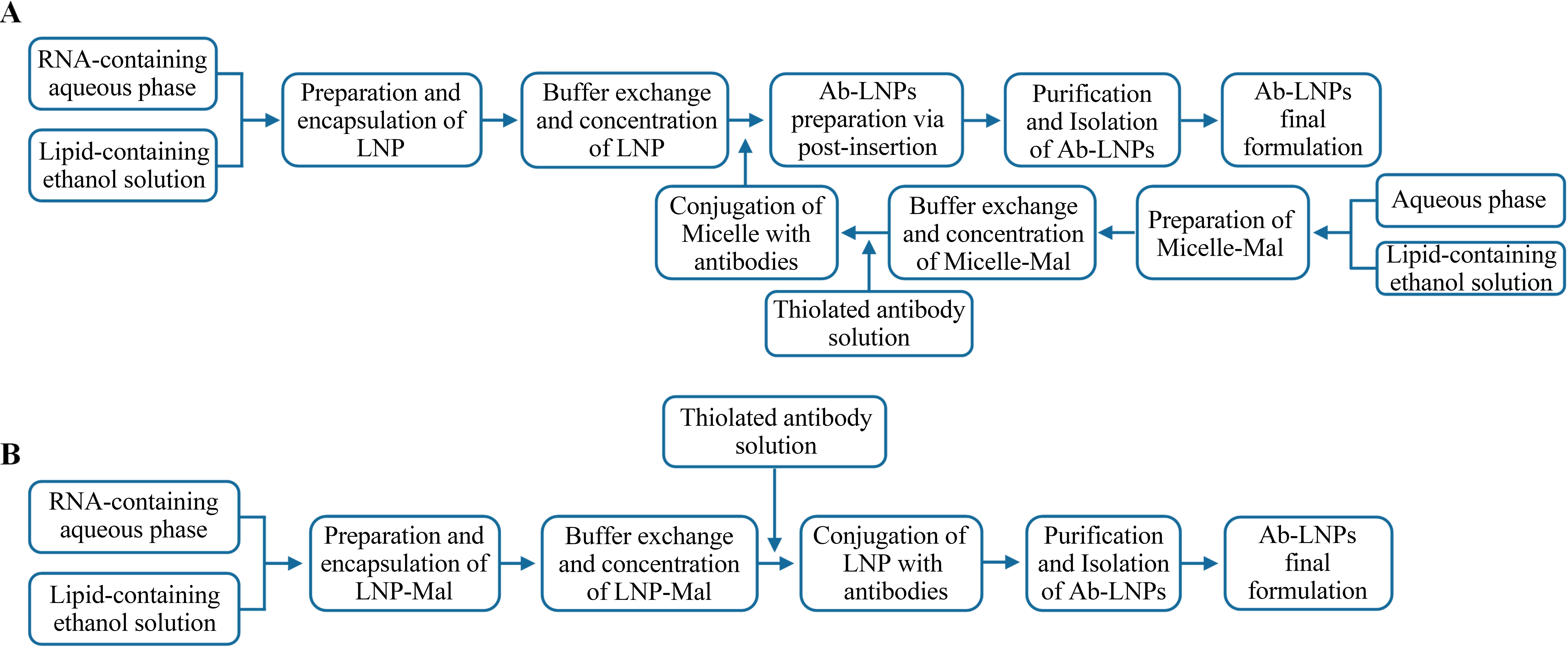
Figure 1. Schematic illustration of the two conjugation modification procedures. (A) Post-insertion; (B) Direct surface conjugation strategies. Conjugation strategies based on thiol chemistry. Ab-LNPs: antibody-targeted lipid nanoparticles.
From a CMC perspective, both methods have their own advantages and disadvantages (Table 1). The post-insertion method isolates the conjugation reaction from LNP formation, minimizing potential impacts on mRNA encapsulation within the LNPs. Additionally, by controlling and purifying intermediates such as antibody micelles, the number of antibodies inserted can be effectively managed, leading to good batch-to-batch consistency. However, due to the need for intermediate preparation, this method presents greater challenges during scale-up production, requiring extensive QC measures. Furthermore, fusion efficiency relies on spontaneous thermodynamic processes, which can lead to unpredictable insertion levels. In comparison, direct surface conjugation involves only an additional conjugation step, simplifying the process and facilitating scale-up production. Nevertheless, the conjugation reaction takes place in the presence of LNPs, which may affect the stability of the encapsulated mRNA. Additionally, conjugation efficiency can be influenced by reaction conditions and may be difficult to control.
| Dimensions | Conjugation strategies | |
| Post-insertion | Direct surface conjugation | |
| Process complexity | High (Multiple steps, requires intermediate preparation) | Low (Adds only a conjugation reaction step) |
| Batch-to-batch consistency | High (Conjugation density controllable via intermediate feeding) | Medium (Conjugation efficiency itself can be less controllable) |
| Impact on encapsulated nucleic acid | Low (Conjugation reaction is performed independently) | Medium (Conjugation occurs in the presence of pre-formed LNPs) |
| Reliability of conjugation process | Low to medium (Stochastic insertion may fail) | High (Covalent reaction is deterministic) |
| Scalability & manufacturing difficulty | High (Challenging intermediate quality control; variable fusion efficiency) | Medium (Simpler process; covalent bonds resist dissociation) |
CMC: chemistry, manufacturing, and controls; LNPs: lipid nanoparticles.
Regardless of the conjugation approach, antibody modification is an essential step. Besides thiol-based chemistry[17], click chemistry, such as alkyne-azide reactions[18], is also commonly used, and the impact of the conjugation chemistry impact on Ab-LNPs may vary considerably. Beyond conjugation methods, Ab-LNPs face additional challenges including robust scale-up, minimization of aggregation, and QC.
3. Antibody Modification Approaches
Antibody modification approaches have evolved from random conjugation to precise site-specific modification using mild aqueous reactions[19,20]. Traditional modification strategies primarily rely on the non-site-specific conjugation of the ε-amino group on lysine (Lys) residues. This is typically achieved through one of two common routes: either via direct reaction using reagents such as
To overcome the limitations of non-specific conjugation, site-specific strategies have emerged. Among these, cysteine (Cys)-based thiol chemistry and azide-alkyne cycloaddition click chemistry are two mainstream approaches, exhibiting fundamental differences in stoichiometric control and product uniformity. Early thiol chemistry predominantly employed maleimide reagents, which react with thiols with high selectivity and rapid kinetics (second-order rate constants up to 102-104 M-1·s-1) under physiological
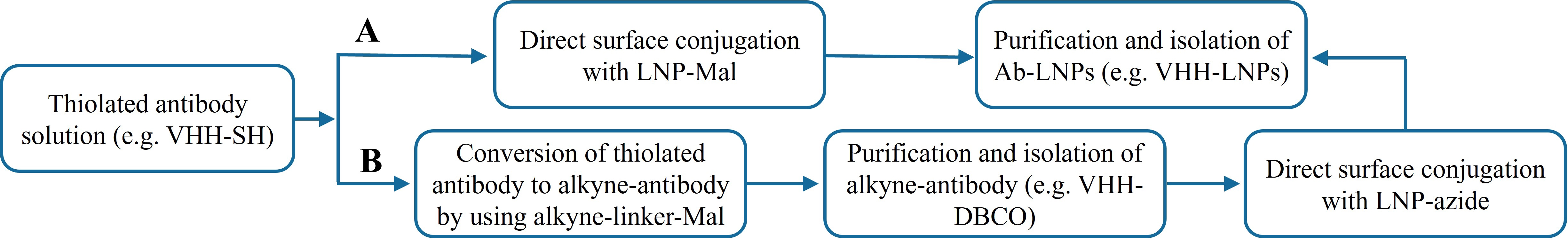
Figure 2. Schematic illustration of the two site-specific conjugation procedures. (A) Thiol chemistry; (B) Click chemistry. VHH: variable domain of heavy-chain antibodies;
As exemplified by VHH (nanobodies), a class of single-domain antibodies increasingly employed in pharmaceutical development and targeted delivery systems, these proteins are typically produced via expression in systems such as Escherichia coli (E.coli) or Chinese hamster ovary (CHO) cells[31-35]. Among these, the E.coli system offers distinct practical advantages, including significantly shorter production cycles and lower costs, compared with the CHO platform[36,37]. Regardless of the expression system used, the resulting VHH requires subsequent functionalization through chemical conjugation. Comparative studies have shown that biotinylated VHH produced via click chemistry exhibits a biotin-to-VHH molar ratio close to the ideal range of 0.6-0.8:1. In contrast, the same VHH conjugated via thiol-maleimide chemistry shows a significantly higher ratio of 1.3-1.8:1, confirming the generation of multi-modified products. Functionally, while both methods generally preserve antigen-binding capability, click chemistry often better maintains or even enhances binding affinity due to its defined linkage site, minimal steric interference, and high product homogeneity, advantages particularly evident when conjugating large molecules like PEG.
However, research indicates that the conjugation chemistry itself can profoundly and unexpectedly influence biological behavior. Both the most popular conjugation chemistries, thiol-maleimide and DBCO-azide click chemistry, can significantly activate the complement system, albeit through distinct mechanisms (Figure 3). For DBCO-azide chemistry, the issue stems from the hydrophobicity of the DBCO moiety[38]. Studies demonstrate that DBCO-modified antibodies can aggregate on nanoparticle surfaces, and these surface protein aggregates become potent activators of the classical complement pathway. In mouse inflammatory models, this leads to abnormal accumulation of antibody-liposome complexes in the lungs (up to 210% ID/g), an effect completely abolished in complement C3 knockout mice, confirming the critical role of complement activation[18]. Notably, this aggregation phenomenon occurs specifically at the nanoparticle interface and is not observed in solution, highlighting the unique effects of the nano-bio interface. For thiol-maleimide chemistry, the mechanism of complement activation is different. The problem lies not with the thiol moiety, but with residual free maleimide groups. These highly reactive groups can undergo non-specific conjugation with thiol-rich plasma proteins, primarily albumin. When albumin “decorates” the nanoparticle surface, it creates sites for attack by the alternative complement pathway. Increasing the molar percentage of maleimide-PEG lipids in liposomes directly leads to a dose-dependent increase in the concentration of the complement activation product C3a, an effect not observed with azide groups. This complement activation not only alters biodistribution but also induces significant toxicities, including a 50% decrease in platelet count and increased hematocrit, which are typical manifestations of complement activation-related pseudoallergy. These findings suggest that “bioorthogonality” in the chemical sense (no side reactions with biological molecules) may differ from its meaning in the immunological sense (no activation of the innate immune system). While the DBCO-azide reaction is chemically specific and orthogonal, its hydrophobic nature triggers undesirable biological responses. To address these issues, in click chemistry,
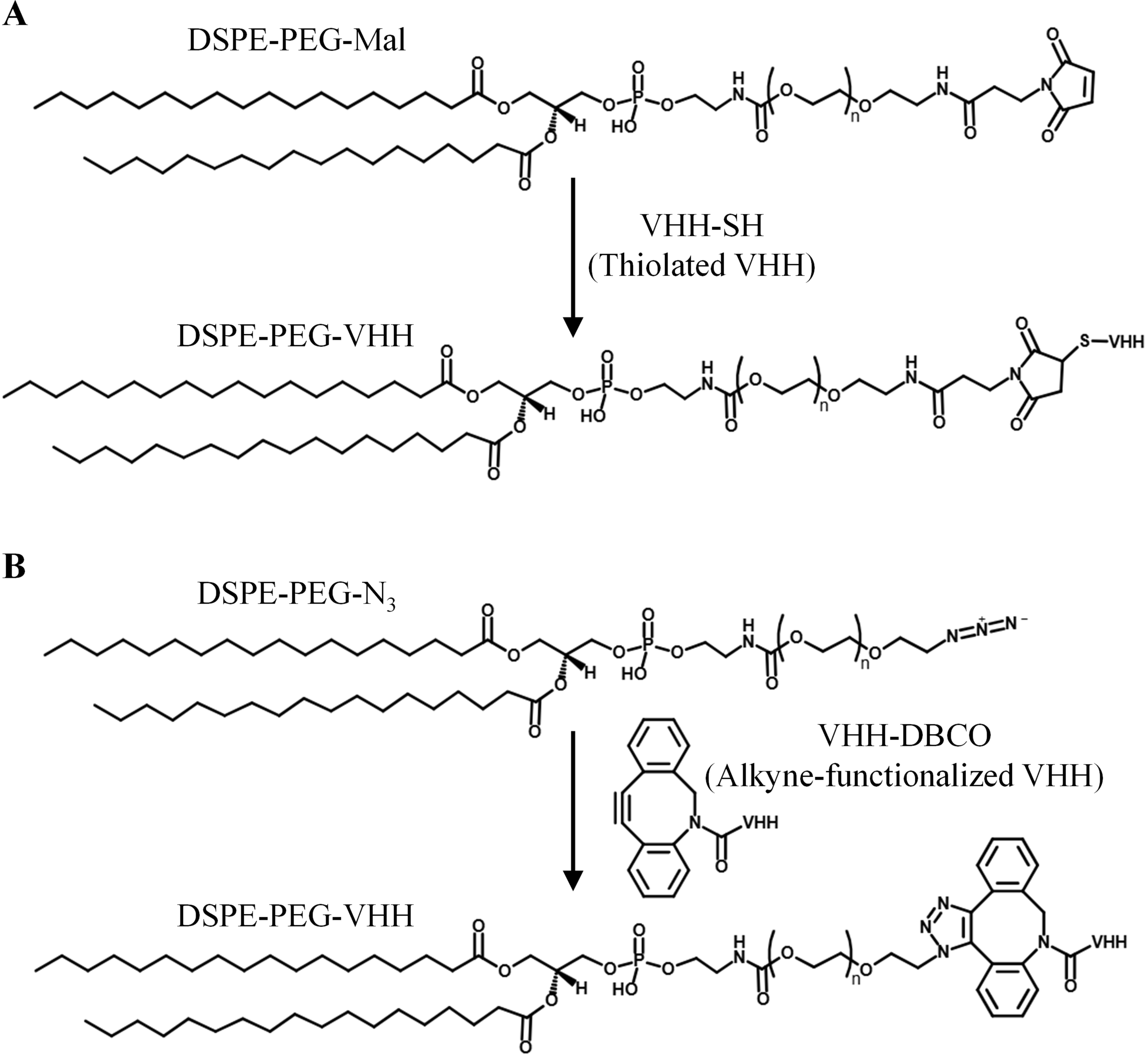
Figure 3. Schematic illustration of conjugation strategies for VHH. (A) Thiol-maleimide reaction; (B) Azide-alkyne click chemistry. Created in InDraw. VHH: variable domain of heavy-chain antibodies.
Click chemistry generally offers advantages in conjugation efficiency over thiol chemistry, and optimization of the conjugation process can also significantly enhance efficiency. For instance, maleimide activity decreases by approximately 10% after 7 days of storage at 4 °C, but by up to 40% when stored at 20 °C for the same duration. This indicates that optimizing conjugation temperature and duration can improve the efficiency of thiol chemistry. Similarly, for different types of protein antibodies, optimization of reaction stoichiometry can enhance conjugation efficiency. For example, in the conjugation of the small peptide cRGDfK with maleimide, the optimal maleimide-to-thiol molar ratio was 2:1, achieving 84% efficiency, while for the larger nanobody 11A4, the optimal ratio was 5:1, with 58% efficiency. Ratios below these optima lead to significantly reduced efficiency, while excessive ligand amounts cause waste without increasing surface loading, suggesting a steric saturation effect, where larger antibody molecules require more maleimide reaction sites. It is noteworthy that introducing site-specific Cys residues at antibody termini
In summary, antibody modification has evolved from simple sequence-based synthesis into an interdisciplinary field integrating protein engineering, materials science, immunology, and pharmacology. The choice of modification strategy for antibody conjugation onto lipid nanoparticles is no longer solely focused on reaction rate and specificity. It now requires a careful balance, considering the overall system stability, scalability, and potential biological consequences.
4. The Hurdles of The Scale-Up Process
The scale-up production of Ab-LNPs fundamentally revolves around resolving the core conflict between translating the highly controlled, precision manual operations, essentially “customized chemistry”, in the laboratory, into a robust, uniform, and reproducible “continuous production process” within large-scale equipment. Beyond the universal scaling challenges shared with conventional LNPs, such as mixing and purification, Ab-LNPs present a series of unique, interrelated, and complex difficulties inherent to their specific antibody conjugation and subsequent antibody purification steps during scale-up. The most immediate manifestation is that, as batch sizes increase, the efficiency of successfully linking target antibodies to the lipid nanoparticle surface (conjugation efficiency) often decreases rather than increases. Furthermore, the proportion of free antibodies, aggregates, and even visible precipitates in the post-reaction system significantly rises, leading to low yield of the active ingredient and inconsistent product quality[40,41]. In actual scale-up production, aggregation must be avoided, while ensuring that the conjugation efficiency loss remains within 5-15%. These issues are not isolated events, but stem from fundamental changes in the physicochemical environment during scale-up. These changes include uneven mixing, decreased mass transfer efficiency, differences in shear force distribution, and accumulation of side reactions due to prolonged reaction times. These factors collectively cause process failure upon
In laboratory research on Ab-LNPs, the pursuit often focuses on the ultimate target affinity of the modified antibody or the highest
During the scale-up of the conjugation process, a key challenge is reproducing the “instantaneous” and “uniform” reaction conditions of small-scale operations in large-scale reactors[40]. In the laboratory, using pipettes or micro-mixers, antibody/reducing agent solutions can achieve molecular-level homogeneous mixing within milliseconds, ensuring each antibody molecule experiences an almost identical, mild reduction environment to expose a predetermined number of thiol groups. However, in reactors of several liters or larger, mixing achieved by top-stirring is macroscopic and relatively slow, inevitably creating concentration and temperature gradients. Localized areas may temporarily experience excessively high reducing agent concentrations, leading to
The scale-up of Ab-LNPs purification is another intricate battle of fine separation within a complex mixture[41]. The post-reaction crude product is a “cocktail” of coexisting phases, containing the target Ab-LNPs, unreacted free antibodies, excess antibody-lipid conjugates (for post-insertion), chemical by-products, and aggregates of various sizes and compositions. The difficulty in purification scale-up lies in the fact that these impurities are often very similar in physicochemical properties to the target product
While deeply optimizing process scale-up, it is crucial to attach high importance to the long-term stability of Ab-LNPs, which directly impacts product shelf life and clinical efficacy. As mentioned, while widely used, traditional maleimide-thiol chemistry may undergo retro-Michael addition or exchange with serum thiols in vivo/in vitro, leading to antibody detachment and loss of targeting
In summary, the process scale-up of Ab-LNPs is a multi-dimensional challenge. It requires moving beyond the optimization of individual technical points to establish a holistic, systemic capability spanning “molecular design-process development-production realization”. By selecting robust antibody candidates at the design source, gaining a deep understanding and precisely controlling the scaled-up physicochemical microenvironment during process development, pursuing a balance between high resolution and product-friendliness in purification strategies, and simultaneously addressing the long-term stability of conjugation chemistry and formulation systems, can the immense potential of this technology be transformed into stable, reliable, and affordable industrial products. This is the path to truly bridging scientific innovation to benefit patients broadly. Moreover, an increased antibody conjugation density does not necessarily correlate with enhanced targeting efficacy. Excessive surface modification may, in fact, impair delivery efficiency, potentially through the promotion of protein corona formation or the introduction of steric hindrance that limits receptor accessibility. Therefore, the optimal antibody-to-lipid ratio should be empirically optimized via systematic in vitro and in vivo evaluations.
5. QC Considerations
In the scale-up production process of Ab-LNPs, the establishment and implementation of a QC system are core to ensuring product consistency, safety, and efficacy. As a type of functionalized nano-delivery system, Ab-LNPs have a structural complexity far exceeding that of traditional LNPs, mainly reflected in the additional QC dimensions and process challenges introduced by antibody modification. As noted in the literature, comprehensive characterization of complex nano-carriers requires multiple complementary, label-free biophysical techniques (such as dynamic light scattering (DLS), multi-angle DLS, electrophoretic light scattering (ELS), nanoparticle tracking analysis (NTA), SEC combined with static light scattering, and differential scanning calorimetry (DSC), etc.) to obtain CQA, including size, distribution, concentration, charge, and thermal stability[47,48]. Although Ab-LNPs share common basic QC indicators such as size, distribution, and encapsulation efficiency with traditional LNPs, their unique antibody modification features significantly increase process complexity and QC difficulty. As the key functional component for targeting, the efficiency, stability, activity, and residual levels of antibody modifications directly determine the targeting efficacy and safety of Ab-LNPs[47]. Therefore, during scale-up, systematic and sensitive monitoring strategies must be established to guide the optimization and standardization of conjugation processes, ensuring batch-to-batch consistency and feasibility for clinical translation (Table 2).
| Key attribute | Description |
| Antibody surface density | Average number of antibody molecules conjugated per particle surface; impacts targeting efficiency and in vivo behavior |
| Conjugation efficiency | Extent of the chemical conjugation reaction, monitored to ensure process control |
| Free antibody removal | Removal of unconjugated free antibodies to prevent competitive inhibition or immune responses |
| Residual free antibody | Quantification of any remaining free antibodies in the final product |
| Binding activity | Assessment of the ability of conjugated antibodies to bind to the target antigen |
| Structural stability | Evaluation of thermal stability and aggregation propensity of the Ab-LNP construct |
| Immunogenicity risk | Assessment of potential new immunogenic risks from the conjugation process |
| Particle size | Hydrodynamic diameter of the particles. |
| Surface charge | Net surface charge influencing stability and biodistribution. |
| Encapsulation efficiency | Proportion of total drug successfully encapsulated within the particles. |
| Drug loading | Absolute amount of drug per particle or unit mass. |
| Polydispersity index | Measure of the width of particle size distribution, indicating batch homogeneity. |
| Lipid identity | Confirmation of the types of lipids present. |
| Lipid concentration | Quantification of lipid amounts for batch consistency. |
| Sterility | Ensuring the product is free from viable microorganisms. |
| Endotoxin level | Ensuring the product is below acceptable endotoxin limits. |
Ab-LNPs: antibody-targeted lipid nanoparticles.
Firstly, monitoring the antibody modification process is the primary QC point in Ab-LNP process scale-up. The conjugation of antibodies to lipid nanoparticles is not a simple physical mixture but involves multiple factors such as chemical bond formation, spatial orientation control, and density optimization. Slight fluctuations in any process parameter may lead to variations in conjugation efficiency, antibody conformation, or surface distribution, thereby affecting targeting performance. During scale-up, it is necessary to monitor key parameters of the conjugation reaction in real-time, such as reaction time, temperature, pH, and material ratios, and to ensure controllability of the conjugation step by establishing appropriate intermediate testing methods (e.g., purity and concentration analysis of antibody-lipid conjugates). For example, ultra-high-performance liquid chromatography coupled with charged aerosol detection (CAD), high-performance liquid chromatography (HPLC) with evaporative light scattering detection, or HPLC-CAD can be used for the quantitative analysis of functionalized lipids and antibody-lipid conjugates (in post-insertion methods), ensuring accurate material input and monitoring of reaction progress[49]. Furthermore, by optimizing the eluent pH and sample pretreatment diluent, baseline separation and accurate quantification of multi-component lipids can be achieved, providing stable guidance for monitoring lipid degradation and impurities.
Additionally, antibody density after Ab-LNP conjugation is a key metric for evaluating modification effectiveness. Excessively high or low antibody density may both affect targeting efficiency and in vivo behavior. Methods such as quantitative SDS-PAGE combined with silver staining, surface plasmon resonance (SPR), biolayer interferometry (BLI), enzyme-linked immunosorbent assay (ELISA), or nano-flow cytometry can be used to determine antibody density, providing data support for process optimization[50]. Particularly, SPR and BLI technologies can monitor the binding kinetics between antibodies and target receptors in real-time, not only for density assessment but also indirectly reflecting the functional status of antibodies post-conjugation. SDS-PAGE with silver staining has been successfully used to quantify the surface modification amounts of antibodies such as Herceptin on lipid nanoparticles, and its bioactivity was validated through antibody-dependent cell-mediated cytotoxicity, demonstrating the reliability and practicality of this method, and providing a feasible path for standardized antibody density analysis of Ab-LNPs[12,51,52].
Secondly, the removal of free antibodies is a critical step in the purification process of Ab-LNPs and must be included as a key item in the QC system. Unconjugated free antibodies remaining in the final product can not only competitively inhibit the targeted binding of Ab-LNPs, but may also cause unintended immune responses or pharmacokinetic interference. Therefore, after the conjugation reaction, free antibodies must be effectively removed through methods such as dialysis, ultrafiltration, or chromatography, and sensitive, specific detection methods must be established for quantitative control of residual levels[50]. Commonly used detection methods include SEC, HPLC coupled with ultraviolet or fluorescence detection, and ELISA based on antigen-antibody interactions. Among these, SEC can separate free antibodies from Ab-LNPs based on molecular weight differences, but care must be taken to avoid nanoparticle adsorption or aggregation in the column[47]. Literature indicates that SEC coupled with UV, multi-angle light scattering (MALS), and refractive index detectors successfully achieves high-resolution analysis of LNP size distribution, molecular weight distribution, and siRNA loading, demonstrating the powerful separation and quantification capabilities of SEC combined with multiple detectors in complex nano-systems, and providing technical reference for the separation and detection of free antibodies in
Furthermore, whether the antibodies retain their structural and functional integrity post-modification is a core element in evaluating the quality of Ab-LNPs. The conjugation process may cause antibody conformational changes, aggregation, degradation, or masking of active sites, leading to loss of targeting function or increased immunogenicity. Therefore, a series of biophysical and functional testing methods must be established to comprehensively assess the activity, stability, and immunogenicity of antibodies after modification. For binding activity, methods such as SPR, BLI, or flow cytometry can be used to measure the affinity and binding specificity of Ab-LNPs to target antigens, ensuring that conjugation does not affect the spatial conformation of the antibody’s complementarity-determining regions[50,53]. Stability assessment involves thermal stability (via DSC or differential scanning fluorimetry), aggregation propensity (via DLS, SEC, or MALS), and storage stability (monitoring antibody shedding or degradation under different temperature and pH conditions). The literature, by combining DSC and DLS thermal scans, revealed different aggregation and structural transition behaviors of empty and full rAAV5 under thermal stress, emphasizing the importance of orthogonal techniques for understanding the stability of complex nano-carriers[54]. Additionally, conjugation may expose new epitopes or alter original immunogenicity profiles, requiring assessment of potential immune risks through methods such as
The establishment and validation of QC methods are the “eyes” of process scale-up, and their accuracy, precision, and robustness directly determine the understanding and control of the process. As a complex nano-drug product, QC for Ab-LNPs spans multiple disciplines, covering physical chemistry, biology, and immunology. For example, size and size distribution can be characterized by DLS, NTA, or field-flow fractionation coupled with multi-angle light scattering[54]. The latter is particularly suitable for high-resolution analysis of polydisperse systems and monitoring size changes in biological media, as literature notes that AF4-MALS-DLS can detect subtle differences between LNP batches and changes in size distribution after serum incubation compared to batch-mode DLS, providing a high-resolution tool for predicting the behavior of Ab-LNPs in complex biological environments[48]. Surface charge
In the context of industrial-scale production, QC strategies must also consider the high-throughput, automation, and online monitoring potential of methods to meet the demands of large-scale manufacturing. Simultaneously, the application of the Quality by Design concept is crucial. By identifying CMA, CPP, and CQA, a design space and control strategy can be established to ensure the robustness of product quality during scale-up. Regulatory agencies such as the FDA have issued guidelines for nano-drugs, emphasizing the importance of process controllability and product characterization[43]. Therefore, the QC system for Ab-LNPs must be comprehensive, systematic, and compliant with regulatory requirements.
In summary, the QC of Ab-LNPs during scale-up is a multi-dimensional systems engineering effort, with its uniqueness primarily stemming from the introduction of antibody modifications. From monitoring the conjugation process and removing free antibodies to verifying antibody functional integrity, each step relies on advanced, sensitive, and well-validated QC methods. These methods often need to draw from and integrate mature characterization strategies in fields such as viral vectors and traditional LNPs, combined with specific analytical tools from antibody engineering. These methods are applied not only in process development and optimization, but also serve as the cornerstone for ensuring product batch-to-batch consistency, safety, and efficacy.
In conclusion, whether using post-insertion or surface modification approaches to prepare Ab-LNPs, it is necessary to evaluate the antibody modification methods, ensure the robustness of unique processes such as conjugation and purification steps, and continuously optimize the analytical methods for key QC items, paving the way for the successful industrialization of this technology.
Authors contribution
Huang R: writing-original draft, writing-review & editing, visualization.
Wang Y, Shi T, Deng C, Xu C: writing-review & editing.
Piao X, Ren K: Supervision, conceptualization, writing-review & editing.
Conflicts of interest
Congcong Xu is a Junior Executive Editor of BME Horizon. Ri Huang, Yifan Wang, Xijun Piao and Keyun Ren are affiliated with CATUG Biotechnology. The other authors declare no conflicts of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
Not applicable.
Funding
None.
Copyright
© The Author(s) 2026.
References
-
1. Parhiz H, Atochina-Vasserman EN, Weissman D. mRNA-based therapeutics: Looking beyond COVID-19 vaccines. Lancet. 2024;403:1192-1204.[DOI]
-
2. Verbeke R, Hogan MJ, Loré K, Pardi N. Innate immune mechanisms of mRNA vaccines. Immunity. 2022;55(11):1993-2005.[DOI]
-
3. Cullis PR, Felgner PL. The 60-year evolution of lipid nanoparticles for nucleic acid delivery. Nat Rev Drug Discov. 2024;23(9):709-722.[DOI]
-
4. Zong Y, Lin Y, Wei T, Cheng Q. Lipid nanoparticle (LNP) enables mRNA delivery for cancer therapy. Adv Mater. 2023;35(51):2303261.[DOI]
-
5. Breda L, Papp TE, Triebwasser MP, Yadegari A, Fedorky MT, Tanaka N, et al. In vivo hematopoietic stem cell modification by mRNA delivery. Science. 2023;381(6656):436-443.[DOI]
-
6. Bot A, Scharenberg A, Friedman K, Guey L, Hofmeister R, Andorko JI, et al. In vivo chimeric antigen receptor (CAR)-T cell therapy. Nat Rev Drug Discov. 2026;25(2):116-137.[DOI]
-
7. Villanueva MT. In vivo engineering broadens CAR-T cell therapy use. Nat Rev Drug Discov. 2025;24(9):664.[DOI]
-
8. Park W, Choi J, Hwang J, Kim S, Kim Y, Shim MK, et al. Apolipoprotein fusion enables spontaneous functionalization of mRNA lipid nanoparticles with antibody for targeted cancer therapy. ACS Nano. 2025;19(6):6412-6425.[DOI]
-
9. Bimbo JF, van Diest E, Murphy DE, Ashoti A, Evers MJW, Narayanavari SA, et al. T cell-specific non-viral DNA delivery and in vivo CAR-T generation using targeted lipid nanoparticles. J Immunother Cancer. 2025;13(7):e011759.[DOI]
-
10. Wang S, Wang H, Drabek A, Smith WS, Liang F, Huang ZR. Unleashing the potential: Designing antibody-targeted lipid nanoparticles for industrial applications with CMC considerations and clinical outlook. Mol Pharm. 2024;21(1):4-17.[DOI]
-
11. van Moorsel MVA, Urbanus RT, Verhoef S, Koekman CA, Vink M, Vermonden T, et al. A head-to-head comparison of conjugation methods for VHHs: Random maleimide-thiol coupling versus controlled click chemistry. Int J Pharm X. 2019;1:100020.[DOI]
-
12. Lin Y, Cheng Q, Wei T. Surface engineering of lipid nanoparticles: Targeted nucleic acid delivery and beyond. Biophys Rep. 2023;9(5):255-278.[DOI]
-
13. Rurik JG, Tombácz I, Yadegari A, Méndez Fernández PO, Shewale SV, Li L, et al. CAR T cells produced in vivo to treat cardiac injury. Science. 2022;375(6576):91-96.[DOI]
-
14. Hunter TL, Bao Y, Zhang Y, Matsuda D, Riener R, Wang A, et al. In vivo CAR T cell generation to treat cancer and autoimmune disease. Science. 2025;388(6753):1311-1317.[DOI]
-
15. Marques AC, Costa PJ, Velho S, Amaral MH. Functionalizing nanoparticles with cancer-targeting antibodies: A comparison of strategies. J Control Release. 2020;320:180-200.[DOI]
-
16. Tombácz I, Laczkó D, Shahnawaz H, Muramatsu H, Natesan A, Yadegari A, et al. Highly efficient CD4+ T cell targeting and genetic recombination using engineered CD4+ cell-homing mRNA-LNPs. Mol Ther. 2021;29(11):3293-3304.[DOI]
-
17. Parayath NN, Stephan SB, Koehne AL, Nelson PS, Stephan MT. In vitro -transcribed antigen receptor mRNA nanocarriers for transient expression in circulating T cells in vivo. Nat Commun. 2020;11:6080.[DOI]
-
18. Zaleski MH, Chase LS, Hood ED, Wang Z, Nong J, Espy CL, et al. Conjugation chemistry markedly impacts toxicity and biodistribution of targeted nanoparticles, mediated by complement activation. Adv Mater. 2025;37(5):2409945.[DOI]
-
19. Oude Blenke E, Klaasse G, Merten H, Plückthun A, Mastrobattista E, Martin NI. Liposome functionalization with copper-free “click chemistry”. J Control Release. 2015;202:14-20.[DOI]
-
20. Rahman MM, Wang J, Wang G, Su Z, Li Y, Chen Y, et al. Chimeric nanobody-decorated liposomes by self-assembly. Nat Nanotechnol. 2024;19(6):818-824.[DOI]
-
21. Junutula JR, Raab H, Clark S, Bhakta S, Leipold DD, Weir S, et al. Site-specific conjugation of a cytotoxic drug to an antibody improves the therapeutic index. Nat Biotechnol. 2008;26(8):925-932.[DOI]
-
22. Gordon MR, Canakci M, Li L, Zhuang J, Osborne B, Thayumanavan S. Field guide to challenges and opportunities in antibody–drug conjugates for chemists. Bioconjug Chem. 2015;26(11):2198-2215.[DOI]
-
23. Wu KL, Yu C, Lee C, Zuo C, Ball ZT, Xiao H. Precision modification of native antibodies. Bioconjug Chem. 2021;32(9):1947-1959.[DOI]
-
24. Chen MZ, Yuen D, McLeod VM, Yong KW, Smyth CH, Herling BR, et al. A versatile antibody capture system drives specific in vivo delivery of mRNA-loaded lipid nanoparticles. Nat Nanotechnol. 2025;20(9):1273-1284.[DOI]
-
25. Alradwan IA, Alnefaie MK, AL Fayez N, Aodah AH, Majrashi MA, Alturki M, et al. Strategic and chemical advances in antibody–drug conjugates. Pharmaceutics. 2025;17(9):1164.[DOI]
-
26. Luu T, Gristwood K, Knight JC, Jörg M. Click chemistry: Reaction rates and their suitability for biomedical applications. Bioconjug Chem. 2024;35(6):715-731.[DOI]
-
27. Martínez-Jothar L, Doulkeridou S, Schiffelers RM, Torano JS, Oliveira S, van Nostrum CF, et al. Insights into maleimide-thiol conjugation chemistry: Conditions for efficient surface functionalization of nanoparticles for receptor targeting. J Control Release. 2018;282:101-109.[DOI]
-
28. Wang Y, Xie F, Liu L, Xu X, Fan S, Zhong W, et al. Development of applicable thiol-linked antibody–drug conjugates with improved stability and therapeutic index. Drug Deliv. 2022;29(1):754-766.[DOI]
-
29. Ochtrop P, Hackenberger CPR. Recent advances of thiol-selective bioconjugation reactions. Curr Opin Chem Biol. 2020;58:28-36.[DOI]
-
30. Chudasama V, Maruani A. A little less conjugation, a little more accuracy. Nat Chem. 2016;8(2):91.[DOI]
-
31. Haueis L, Stech M, Kubick S. A cell-free expression pipeline for the generation and functional characterization of nanobodies. Front Bioeng Biotechnol. 2022;10:896763.[DOI]
-
32. Verma V, Sinha N, Raja A. Nanoscale warriors against viral invaders: A comprehensive review of Nanobodies as potential antiviral therapeutics. mAbs. 2025;17(1):2486390.[DOI]
-
33. Wu Y. Trends in nanobody technology in industrialization. Discov Nano. 2025;20:23.[DOI]
-
34. Walsh G, Walsh E. Biopharmaceutical benchmarks 2022. Nat Biotechnol. 2022;40(12):1722-1760.[DOI]
-
35. Mullin M, McClory J, Haynes W, Grace J, Robertson N, van Heeke G. Applications and challenges in designing VHH-based bispecific antibodies: Leveraging machine learning solutions. mAbs. 2024;16:2341443.[DOI]
-
36. İncir İ, Kaplan Ö. Escherichia coli in the production of biopharmaceuticals. Biotechnol Appl Biochem. 2025;72(2):528-541.[DOI]
-
37. Blueggel M, Gül D, Stauber RH, Knauer SK. Small but mighty: The versatility of nanobodies in modern medicine. Nanoscale Horiz. 2025;10(10):2158-2171.[DOI]
-
38. Notabi MK, Arnspang EC, Andersen MØ. Antibody conjugated lipid nanoparticles as a targeted drug delivery system for hydrophobic pharmaceuticals. Eur J Pharm Sci. 2021;161:105777.[DOI]
-
39. Peciak K, Laurine E, Tommasi R, Choi JW, Brocchini S. Site-selective protein conjugation at histidine. Chem Sci. 2019;10(2):427-439.[DOI]
-
40. Wicki A, Ritschard R, Loesch U, Deuster S, Rochlitz C, Mamot C. Large-scale manufacturing of GMP-compliant anti-EGFR targeted nanocarriers: Production of doxorubicin-loaded anti-EGFR-immunoliposomes for a first-in-man clinical trial. Int J Pharm. 2015;484:8-15.[DOI]
-
41. Nellis DF, Ekstrom DL, Kirpotin DB, Zhu J, Andersson R, Broadt TL, et al. Preclinical manufacture of an anti-HER2 scFv-PEG-DSPE, liposome-inserting conjugate. 1. Biotechnol Progress. 2008;21(1):205-220.[DOI]
-
42. Huang ZR, Tipparaju SK, Kirpotin DB, Pien C, Kornaga T, Noble CO, et al. Formulation optimization of an ephrin A2 targeted immunoliposome encapsulating reversibly modified taxane prodrugs. J Control Release. 2019;310:47-57.[DOI]
-
43. Drug products, including biological products, that contain nanomaterials–Guidance for industry [Internet]. Silver Spring: Center for Drug Evaluation and Research; 2022. Available from: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/drug-products-including-biological-products-contain-nanomaterials-guidance-industry
-
44. Geddie ML, Kohli N, Kirpotin DB, Razlog M, Jiao Y, Kornaga T, et al. Improving the developability of an anti-EphA2 single-chain variable fragment for nanoparticle targeting. mAbs. 2017;9(1):58-67.[DOI]
-
45. Fontaine SD, Reid R, Robinson L, Ashley GW, Santi DV. Long-term stabilization of maleimide–thiol conjugates. Bioconjug Chem. 2015;26(1):145-152.[DOI]
-
46. Aldington S, Bonnerjea J. Scale-up of monoclonal antibody purification processes. J Chromatogr B. 2007;848(1):64-78.[DOI]
-
47. Zhang J, Haas RM, Leone AM. Polydispersity characterization of lipid nanoparticles for siRNA delivery using multiple detection size-exclusion chromatography. Anal Chem. 2012;84(14):6088-6096.[DOI]
-
48. Caputo F, Arnould A, Bacia M, Ling WL, Rustique E, Texier I, et al. Measuring particle size distribution by asymmetric flow field flow fractionation: A powerful method for the preclinical characterization of lipid-based nanoparticles. Mol Pharm. 2019;16(2):756-767.[DOI]
-
49. Kinsey C, Lu T, Deiss A, Vuolo K, Klein L, Rustandi RR, et al. Determination of lipid content and stability in lipid nanoparticles using ultra high-performance liquid chromatography in combination with a Corona Charged Aerosol Detector. Electrophoresis. 2022;43:1091-1100.[DOI]
-
50. Yano Y, Rui T, Hamano N, Haruta K, Kobayashi T, Sato M, et al. Development of a concise and reliable method for quantifying the antibody loaded onto lipid nanoparticles modified with Herceptin. J Immunol Meth. 2023;521:113554.[DOI]
-
51. Bérot A, Maniti O, El Alaoui S, Granjon T, El Alaoui M. Generation of anti-epidermal growth factor receptor-2 (HER2) immunoliposomes using microbial transglutaminase (mTG)-mediated site-specific conjugated antibodies. ACS Pharmacol Transl Sci. 2024;7(10):3034-3044.[DOI]
-
52. Yano Y, Hamano N, Haruta K, Kobayashi T, Sato M, Kikkawa Y, et al. Development of an antibody delivery method for cancer treatment by combining ultrasound with therapeutic antibody-modified nanobubbles using Fc-binding polypeptide. Pharmaceutics. 2023;15(1):130.[DOI]
-
53. Kedmi R, Veiga N, Ramishetti S, Goldsmith M, Rosenblum D, Dammes N, et al. A modular platform for targeted RNAi therapeutics. Nat Nanotechnol. 2018;13(3):214-219.[DOI]
-
54. Markova N, Cairns S, Jankevics-Jones H, Kaszuba M, Caputo F, Parot J. Biophysical characterization of viral and lipid-based vectors for vaccines and therapeutics with light scattering and calorimetric techniques. Vaccines. 2022;10(1):49.[DOI]
Copyright
© The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Publisher’s Note
Share And Cite




